Journal of Environmental and Agricultural Sciences (JEAS). Arshad et al., 2020. Volume 22(1): 49-56
Open Access – Research Article
Evaluation of Entomopathogenic Nematodes and Spinosad against Melon Fruit Fly, Bactrocera cucurbitae (Diptera: Tephritidae)
Aqsa Arshad 1, Muhammad Ishaque Mastoi 3, Asim Munawar 1,2,*, Samar Sohail 4, Chengjuan Liang 5, Fazlullah 6 1 Department of Entomology, University of Agriculture Faisalabad, Pakistan 2 Institute of Insect Sciences, College of Agriculture and Biotechnology, Zhejiang University, China 3 Plant Sciences Division, Pakistan Agricultural Research Council Islamabad, Pakistan 4 Institute of Horticulture, University of Agriculture Faisalabad, Pakistan 5 Institute of Applied Ecology, Fujian Agriculture and Forestry University, China 6 Biological Control Container Laboratory, National Agricultural Research Center, Islamabad, Pakistan
Abstract: Fruit flies are among the most important group of pests causing economical losses by damaging fruits worldwide. They have countless economic significance in Pakistan due to their substantial losses. In current research entomopathogenic nematode (EPNs) and bacterial based spinosad was checked against melon fruit fly, Bactrocera cucurbitae under laboratory conditions. Different concentrations of nematodes and spinosad sole and in combination was applied on B. cucurbitae. Bacterial based spinosad was applied by dip method whereas nematodes were applied on the soil surface. Mortality data was checked against larvae, pupae and adults. Single and combine effect of EPNs and spinosad was also checked on fruit fly developmental time. Results showed that spinosad bacterial based concentrations cause maximum mortality against melon fruit fly as compared to nematode. While combine effect of spinosad and nematode mortality was increased gradually with increasing concentrations as compared to alone treatment. Our results conclude that nematode and bacterial based spinosad have a sound base for the effective management of fruit flies species. These results could be helpful for application of nematode and spinosad in Insect Pest Managements Programs.
Keywords: Mortality, nematodes, spinosad, larvae, pupae, adult, development. *Corresponding author: Asim Munawar: amento3762@hotmail.com Cite this article as: Arshad, A., M.I. Mastoi, A. Munawar, S. Sohail and Fazlullah. 2020. Evaluation of entomopathogenic nematodes and spinosad against melon fruit fly, Bactrocera cucurbitae (Diptera: Tephritidae). Journal of Environmental and Agricultural Sciences. 22(1):49-56.1. Introduction Among vegetables bitter gourd, Momordica charantia L. is considered important part of agriculture product in Pakistan. This vegetable is grown worldwide with more importance in Asia like India, Sri Lanka, Bangladesh, and China as medicinal and nutritional vegetable (Rahman and Norton, 2019; Islam et al., 2017). The bitter gourd production is extremely effected by fruit flies species especially by Bactrocera cucurbitae (Dipteran: Tephritidae), that cause huge economic losses in bitter gourd production (Khursheed and Raj, 2019). Fruit flies especially melon fruit fly (B. cucurbitae) has become most serious pest of cucurbits. While it has been reported in many countries like India, Myanmar, Malaysia, Thailand, China, Philippines, East Africa, Guam, Taiwan, Northern Mariana Islands, Solomon Islands, Papua Guinea, as well as in Hawaiian Island (Dominiak and Worsley, 2018; Hadapad et al., 2016).
B. cucurbitae is widely distributed throughout the world with around 81 host plants as well as major pest of cucurbitaceous crops. The losses due to B. cucurbitae vary between 30-100 % based on crops and seasons (Dhillon et al., 2005). Fruit flies have great importance with quarantine point of view, imposing strict regulations in developing countries while this species causes direct losses by infestation as well as indirect losses in export and import commodities and decreases trade value (Badii et al., 2015; Chen and Ye, 2007).
The adults of B. cucurbitae deposit their eggs within fruit pulp by their sharp ovipositor as well as in plant tissues, young plants, seedlings and unopened flowers by making the plant sick and unable to grow well, and in extreme case, leading to death of plants (Kate et al., 2009). Hatched maggots feed on fruit pulp by making zigzag galleries, and promotes bacterial infections and fungal growths resulting in complete deterioration of fruit (Dominiak, 2018). Melon fly oviposition in upper sheath of fruits not only damages the fruits but also deteriorate their quality as well as economic value (Sapkota et al., 2013). This economical important pest imposing serious threats to fruit industry and in trade values throughout the world (Hossain and Khalequzzaman, 2018; Badii et al., 2015; Joomaye et al., 2000).
Successful control of this fruit fly is a challenge because maggots feed inside the fruits and directly not exposed to pesticides or other suitable control techniques (Deguine et al., 2015; Devaraju et al., 2018). Due to these faced problems the trend for control of fruit flies species now been changing to other suitable pest management tactics like use of biological control agents (Joseph et al., 2017; Klungness et al., 2005). Use of microbial agents as bio-pesticides is considered an effective management component for the control of fruit flies species (Quesada et al., 2012).
Another microbes component, entomopathogenic nematodes (EPNs) are also a possible tools for managing fruit flies (Aatif et al., 2019; Gazit et al., 2000). EPNs have been widely practiced for the control of agricultural pests with importance to fruit flies especially B. cucurbitae, Out of all EPNs Heterorhabditidae has achieved better importance (Ahmad et al., 2017; Joseph et al., 2017; Kapranas et al., 2020). EPNs have been practiced reproductively for the control of soil dwelling pests as well as for above ground insect pests in cryptic habitats (Arthers et al., 2004; Jaffuel et al., 2020; Shapiro-Ilan et al., 2012). They have wide host range spectrum, causing mortality of the host within two days with long period efficacy, easy mass rearing and applications as well as considered environmentally safe (Kaya, 1990).
Insecticides especially the group organophosphates still the main components employed widely for the control of fruit flies as lethal baits (Borges et al., 2015). Microbial pesticides spinosyns relatively received more attention as they are less toxic to humans and environmentally safe. Spinosyns are selective for beneficial insects (Galm and Sparks, 2015; Shutze et al., 2018).
Due to emerging issues by fruit flies outbreak current research was focused on biological management of B. cucurbitae by EPNs and spinosad. Mortality as well as effect of both bio-control agent on developmental time against melon fruit fly was also checked in single and in combinations. The result of this research will provide a sound base for effective IPM strategies.
2. Materials and Methods 2.1. Insect Culture Population of B. cucerbitae was maintained in Laboratory. The infected fruits by melon fruit fly were collected from the fields and were placed in the card boxes half filled with sieved and sterilized sand under optimal conditions 28±2 ºC, 65±5% RH and 12:12 (Day: Light) photoperiod. B. cucurbitae larvae were hatched out from fruit pulp, after completing their growth were going down to the ground sand for pupation. Then pupae were placed in the rearing cages till the adult emergence and placed in incubators without lighting under similar conditions of rearing room. All emerging adults were transferred to mating cages equipped with honey, protein, and sugar-water solution 5% in a small petri dish and dry sugar as well as pieces of cotton moistened with water as a source of protein for females and to improve the activity of mating of males. The eggs were collected from oviposition and placed them onto the artificial diet. The fruits after three days were transferred to card boxes, containing sterilized sand, for obtaining the next progeny and then this procedure was used to mass culture of melon fruit fly. 2.2. Nematodes culture For rearing EPNs and promoting their strains nematodes were cultured on matured larvae of wax moth, Galleria mellonella L. as experienced by other researchers (Kaya and Stock, 1997). Killed larvae of wax moth were placed on white traps for the collection of emerging infective juveniles stages (IJs). Collected nematodes were shifted to petri dishes and wrapped with parafilm and stored at 10ºC. 2.3. Experiment bioassay Experiment was subjected to perform in plastic cups (6 cm diameter, 4 cm height) each filled with 35 g sterilized soil. Cup lids were modified with holes so that normal air can pass. This experiment was conducted on 3 life stages of fruit fly i.e. larvae, pupae and adult. Around ten last instar larvae of fruit fly were placed in separate plastic containers while EPNs concentrations were 100 and 150 IJs/cm2. The EPN H. indica applied with the help of pipette (Kamali et al., 2013). After application of EPNs, larvae were placed in containers for settlement after that two concentrations of Spinosad (30 g and 40 g) were applied with glass stirring rod, and these treatments are used alone and combined form. Ten to nine days old pupae and ten adults were used in separate containers while control treatments received water only. Each treatment consisted of five replicates and whole experiment was repeated three times. First mortality data was taken after 7 days of applications in both alone and combined treatments and then taken on a daily basis continuing up to 14 days. Seven days after treatment, soil was sieved and dead insects were counted. Same treatments was applied to check their effect on developmental time against melon fruit fly, B. cucurbitae. The laboratory conditions were maintained at 25±1°C, 62±5 % RH and 16:8 (Light: Dark) photoperiod (Beris et al., 2013). There were no differences resulted in day night temperature. 2.4. Statistical analysis Mortality rate caused by nematodes and spinosad subjected to analyze with One way Analysis of Variance (ANOVA) using analytical software (Statistix 8.1, SPSS 16.0) in Complete Randomized Design (CRD). When results was significant, comparisons among means were made using Tukey test at 5% significance level. Excel sheet (2016) was used to draw tables and concluding data. 3. Results and Discussion Results of research concluded that the applied concentrations of bio-pesticide spinosad and nematode H. indica showed significant effects against pupae, adult and larvae in alone and in combinations against B. cucurbitae. These both bio-control agents H. indica and bacterial based spinosad used in experiment also showed the significant effect on the developmental stages of B. cucurbitae and the table of analysis of variance of mortality and developmental stages data of B. cucurbitae revealed significant differences between different treatments. 3.1. Effect of bio-control agents against larval mortality of B. cucurbitae Significant difference was observed in third instar larval mortality of B. cucurbitae when treated with different concentrations of spinosad and nematode. Spinosad caused higher mortality as compared to H. Indica: as their maximum concentrations (Table 1). The mortality of larvae increased as the concentration of treatments increased. When treatments applied alone I2 (40mg /30g sand) caused maximum mortality (62.59%) followed by I1 (30mg /30g sand) caused maximum mortality (52.59%), which are in agreement with the findings of Boiteau and Noronha (2007). Moreover, direct spray applications killed flies quickly, consistent with the study in other insects comparing topical versus residual toxicities spinosad, which perform as a nicotinic acetylcholine receptors (Brown 2006). While nematodes Hi1 (100 IJS cm-2 ) caused maximum mortality (42.53%) and Hi2 (150 IJS cm-2 ) caused maximum mortality (45.55 %). While the combined application of both agents yield maximum mortality as compared to alone treatments (Table 1). In combined applications maximum mortality was observed against highest concentrations of both agents (85.92 %) followed by (76.12 %), (74.45%) and (67.78 %). Overall more mortality (85.92%) caused in larval stage because it’s more susceptible stage. Fruit flies maggots are fully active and often moves around, which may provide EPNs and spinosad a greater possibility to be exposed without more struggles. Results of larval mortality with EPNs relate with other researchers to their EPNs efficacy to fruit flies (Lezama-Gutierrez et al., 2006), R. indifferens (Yee and Lacey, 2003 and Sirjani et al. 2009) which also showed higher susceptibility of fruit fly larvae. Larval duration increased as concentration of treatment were increased and larval survival percentage decreased, mean larval survival percentage from 82.95% to 32.80%. (Table 4). Table 1. Mean mortality of B. cucurbitae larvae exposed to two different concentrations of H. indica and Spinosad Means sharing different letters are significantly different at 5% interval (P < 0.05, Tukey HSD test); Hi: Heterorhabditis indica; SP: Spinosad.
The stage of insect development has a significant effect on vulnerability to EPNs (Kaya and Hara, 1980). In general, reduction in adult longevity, oviposition, egg hatching, fecundity. Gazit et al. (2000) also reported similar results when checked different nematodes species against C. capitata. Laborda et al. (2003) and Koppler et al. (2004) reported less activity of EPNs against pupae of fruit fly C. capitata and R. cerasi, respectively.
3.2. Effect of bio-control agents against pupal mortality of B. cucurbitae
Significant difference was observed in pupal mortality of B. cucerbitae when treated with different concentrations of spinosad and nematode. When treatments applied alone I2 (40mg /30g sand) caused maximum mortality (54.26 %) followed by I1 (30mg /30g sand) caused maximum mortality (45.74 %), Hi1 (100 IJS cm-2 ) caused maximum mortality (33.15 %) and Hi2 (150 IJS cm-2 ) caused maximum mortality (42.22 %) (Table 2). Hard integument of the prepupae which prevents EPNs penetration through the puparium. According to Barbosa-Negrisoli et al. (2009) pupa stage of A. fraterculus was susceptible to some EPNs species, including S. riobrave and H. bacteriophora. Radhakrishnan and Shanmugam (2017) reported that by increasing the concentrations of H. indica and S. glaseri, mortality were also increased in S. litura. In their study H. indica reported to be more effective under pot culture conditions. While the combined application of both agents yield maximum mortality as compared to alone treatments. Negrisoli et al. (2010) reported that mortality of the pre-pupae of S. frugiperda was significantly different when treated with insecticide and EPNs as compared single applications of EPNs.
Table 2. Mortality of B. cucurbitae pupae exposed to different concentrations of H. indica and Spinosad
Means sharing different letters are significantly different at 5% interval (P < 0.05, Tukey HSD test); Hi: Heterorhabditis indica; SP: Spinosad.
The stage of insect development has a significant effect on vulnerability to EPNs (Kaya and Hara, 1980). In general, reduction in adult longevity, oviposition, egg hatching, fecundity. Gazit et al. (2000) also reported similar results when checked different nematodes species against C. capitata. Laborda et al. (2003) and Koppler et al. (2004) reported less activity of EPNs against pupae of fruit fly C. capitata and R. cerasi, respectively.
3.2. Effect of bio-control agents against pupal mortality of B. cucurbitae
Significant difference was observed in pupal mortality of B. cucerbitae when treated with different concentrations of spinosad and nematode. When treatments applied alone I2 (40mg /30g sand) caused maximum mortality (54.26 %) followed by I1 (30mg /30g sand) caused maximum mortality (45.74 %), Hi1 (100 IJS cm-2 ) caused maximum mortality (33.15 %) and Hi2 (150 IJS cm-2 ) caused maximum mortality (42.22 %) (Table 2). Hard integument of the prepupae which prevents EPNs penetration through the puparium. According to Barbosa-Negrisoli et al. (2009) pupa stage of A. fraterculus was susceptible to some EPNs species, including S. riobrave and H. bacteriophora. Radhakrishnan and Shanmugam (2017) reported that by increasing the concentrations of H. indica and S. glaseri, mortality were also increased in S. litura. In their study H. indica reported to be more effective under pot culture conditions. While the combined application of both agents yield maximum mortality as compared to alone treatments. Negrisoli et al. (2010) reported that mortality of the pre-pupae of S. frugiperda was significantly different when treated with insecticide and EPNs as compared single applications of EPNs.
Table 2. Mortality of B. cucurbitae pupae exposed to different concentrations of H. indica and Spinosad
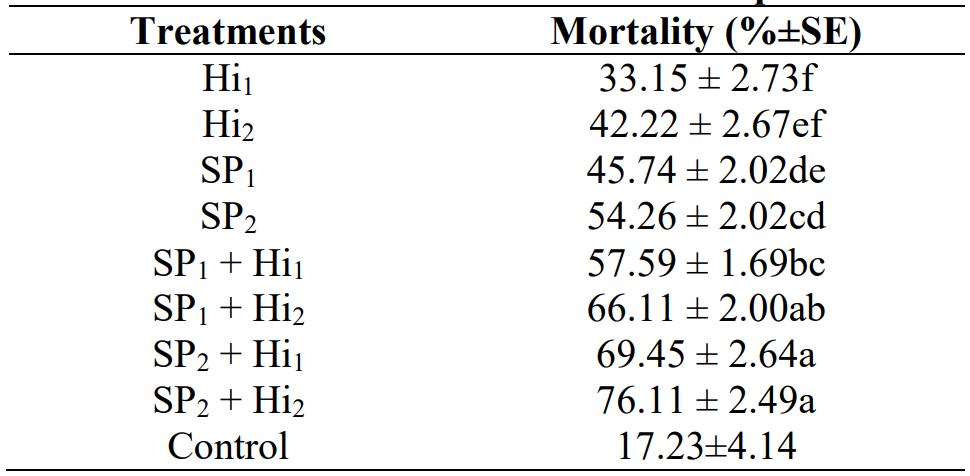 Means sharing different letters within each columns are significantly different at 5 % interval (P < 0.05, Tucky HSD test). Hi1‒ Heterorhabditis indica (100 IJs); Hi2‒ Heterorhabditis indica (150 IJs); SP: Spinosad; S1‒ Spinosad (30 mg); S2‒ Spinosad (40 mg).
3.3. Effect of bio-control agents against adult mortality of B. cucurbitae
The mortality rate of adult fruit flies was increased as the concentration of treatments increased (Table 3). Varying interactions between insecticides and pathogens, like chlorpyrifos have been reported (Koppenhofer and Grewal 2005, Shapiro-Ilan et al. 2011). Alone I2 (40mg /30g sand) caused maximum mortality (52.59 %) followed by I1 (30mg /30g sand; mortality 42.40 %), Hi1 (100 IJs cm-2 ; mortality 30.18 %) and Hi2 (150 IJs cm-2 ; mortality 35.55 %). While the combined application of both agents yield maximum mortality as compared to alone treatments.
Table 3. Mortality of B. cucurbitae larvae exposed to different concentrations of H. indica and Spinosad
Means sharing different letters within each columns are significantly different at 5 % interval (P < 0.05, Tucky HSD test). Hi1‒ Heterorhabditis indica (100 IJs); Hi2‒ Heterorhabditis indica (150 IJs); SP: Spinosad; S1‒ Spinosad (30 mg); S2‒ Spinosad (40 mg).
3.3. Effect of bio-control agents against adult mortality of B. cucurbitae
The mortality rate of adult fruit flies was increased as the concentration of treatments increased (Table 3). Varying interactions between insecticides and pathogens, like chlorpyrifos have been reported (Koppenhofer and Grewal 2005, Shapiro-Ilan et al. 2011). Alone I2 (40mg /30g sand) caused maximum mortality (52.59 %) followed by I1 (30mg /30g sand; mortality 42.40 %), Hi1 (100 IJs cm-2 ; mortality 30.18 %) and Hi2 (150 IJs cm-2 ; mortality 35.55 %). While the combined application of both agents yield maximum mortality as compared to alone treatments.
Table 3. Mortality of B. cucurbitae larvae exposed to different concentrations of H. indica and Spinosad
 Means sharing different letters within each columns are significantly different at 5 % interval (P < 0.05, Tucky HSD test). Hi1‒ Heterorhabditis indica (100 IJs); Hi2‒ Heterorhabditis indica (150 IJs); SP: Spinosad; S1‒ Spinosad (30 mg); S2‒ Spinosad (40 mg).
Results showed that the EPNs species and insecticide concentrations as well as their mixtures had more to be effective on the pupae. In combined application maximum mortality was observed against highest concentrations of both agents (76.11 %) followed by (SP2 + Hi1; 69.45 %), (SP2 + Hi1; 66.11 5%) and (SP1 + Hi1; 57.59 %). All combinations exhibited synergistic interaction. Combine infectivity of H. indica and spinosad presented an additive effect (Table 2). Mean pupal survival percentage ranged from 50.01% (Hi1) to 13.89% (SP2 + Hi2) and in control (74.73%). Guo et al. (2016) showed an additive effect of combinations of H. bacteriophora with imidacloprid and chlorantraniliprole on second instars larvae of H. oblita, causing higher mortality than the nematode species or insecticides alone. High concentrations of the insecticides with nematodes, strongest synergistic effects were found. In combined application maximum mortality was observed against highest concentrations of both agents (SP2 + Hi2; 72.78 %) followed by (SP2 + Hi1; 65.92 %), (SP1 + Hi2; 64.26 %) and (SP1 + Hi1; 54.07 %).
Table 4. Effect of Spinosad and nematode, H. indica on the development of melon fruit fly, B. cucurbitae
Means sharing different letters within each columns are significantly different at 5 % interval (P < 0.05, Tucky HSD test). Hi1‒ Heterorhabditis indica (100 IJs); Hi2‒ Heterorhabditis indica (150 IJs); SP: Spinosad; S1‒ Spinosad (30 mg); S2‒ Spinosad (40 mg).
Results showed that the EPNs species and insecticide concentrations as well as their mixtures had more to be effective on the pupae. In combined application maximum mortality was observed against highest concentrations of both agents (76.11 %) followed by (SP2 + Hi1; 69.45 %), (SP2 + Hi1; 66.11 5%) and (SP1 + Hi1; 57.59 %). All combinations exhibited synergistic interaction. Combine infectivity of H. indica and spinosad presented an additive effect (Table 2). Mean pupal survival percentage ranged from 50.01% (Hi1) to 13.89% (SP2 + Hi2) and in control (74.73%). Guo et al. (2016) showed an additive effect of combinations of H. bacteriophora with imidacloprid and chlorantraniliprole on second instars larvae of H. oblita, causing higher mortality than the nematode species or insecticides alone. High concentrations of the insecticides with nematodes, strongest synergistic effects were found. In combined application maximum mortality was observed against highest concentrations of both agents (SP2 + Hi2; 72.78 %) followed by (SP2 + Hi1; 65.92 %), (SP1 + Hi2; 64.26 %) and (SP1 + Hi1; 54.07 %).
Table 4. Effect of Spinosad and nematode, H. indica on the development of melon fruit fly, B. cucurbitae
 Means sharing different letters within each columns are significantly different at 5 % interval (P < 0.05, Tucky HSD test). Hi1‒ Heterorhabditis indica (100 IJs); Hi2‒ Heterorhabditis indica (150 IJs); SP: Spinosad; S1‒ Spinosad (30 mg); S2‒ Spinosad (40 mg).
According to Zimmerman and Cranshaw (1990) which reported high levels toxicity of chlorpyrifos to S. feltiae but with less toxicity to S. carpocapsae and heterorhabditid. Negrisoli et al. (2010) reported coaction between nematodes (S. glaseri and H. indica) with chlorpyrifos when targeting armyworm, S. frugiperda. According to Jiang and Feng (2006) long term compatibility between two agents (chlorpyrifos and the fungus), were observed as well as synergistic interaction between imidacloprid and white grubs. Koppenhofer et al. (2000) checked that the mechanisms laid to reduced pests activity because of the chemical, which allowed the nematode to overcome the insect’s defenses.
3.4. Effect of bio-control agents against developmental time of B. cucurbitae
For the first time the effect of nematodes and bacterial based spinosad was checked against developmental time of B. cucurbitae. When nematode H. indica and spinosad was applied in single doses the spinosad showed high infection rate which reduced the adult longevity, eggs laying capacity, eggs hatching percentage, fecundity rate, larval duration, pupal duration while nematodes also showed same results with little differences. Pupal survival, larval survival and pre-oviposition period was increased in spinosad treatments, which was observed low in nematodes.
In combine treatments spinosad and nematode with highest concentration (40g, 150IJs) showed gradually highest effect as compared to other treated combinations. While control treated with distilled water showed highest developmental rate. According to current results it was suggested that combination of nematodes and insecticides can provide a best control strategy as compared to others. This was first time study who reported these results, due to this it was becomes difficult to compare with other studies. However we want to compare our results with Guo et al. (2017); Khan et al. (2018) and Heve et al. (2017) who did some aspects of our current research and got significant results as compared to our study. According to reviews, and current research it is suggested that the best approaches to counteract with fruit flies is to use combined treatments of nematodes and bacterial based spinosad, which could provide effective results, and might be helpful at farmer level.
4. Conclusion
Present research was focused to control fruit flies species especially melon fruit fly with appropriate management tactics. In the past researchers largely considered pesticides applications for control of fruit flies, but unfortunately due to multiple application these pests has developed resistance against certain types of organophosphates. Broad spectrum applications of pesticides also imposed some global warming threats including human’s health issues. Under such circumstances current research was focused to control fruit flies species biologically rather than excessive pesticide application. Present research was based on use of EPNs with combination of bacterial based spinosad for successful control of fruit flies. Fruit flies larvae, pupae and adults was exposed to combine and single treatments of both currently applied bio-control agents. Effect of these both bio-control agents was also evaluated on the development of fruit flies. Obtained results suggested that both bio-control agents showed significant control of B. cucurbitae with expected mortalities. Bacterial based spinosad was observed more toxic than EPNs. In combined applications of both biocontrol agents significant results were obtained with causing maximum mortalities. As concentration was increased mortality rate was also increased. This research provides an environmentally safe fruit flies control technique. This research could provide a sound base for the effective management of fruit flies species.
List of Abbreviations: EPNs: Entomopathogenic nematodes, Hi: Heterorhabditis Indica, S: Spinosad, IPM: Integrated Pest Management, RH: Relative Humidity, IJs: Infective Juvenile stage, CRD: Complete Randomized Design, ANOVA: Analysis of Variance, SE: Standard Error.
Competing Interest Statement: This declare that all the authors of this research articles do not have any competing interest.
Author’s Contribution: All the author participate equally in this project. A.A. conducted experiments, while A.M. assisted for data analysis and experiments designs. S.S. assisted practically in experiments as well as in fruit flies collections from the orchards. M.I.M., S.S., C.L. and F. assisted to revise and improvements in the manuscript.
Acknowledgments: Author of this manuscript is thankful to all correspondence authors for help throughout the research during her Master study.
Funding Source: Research project was self-funded without any financial assistance.
References
Ahmad, G., A.A. Khan and S. Ansari. 2017. Interaction of a fly ash and root-knot nematode pathogens on Pumpkin (Cucurbita moschata Duch. ex Lam.). Tro. Plant. Res. 4(3): 449-455.
Akhurst, R and K. Smith. 2002. Regulation and safety. In: Gaugler, R. (Ed.), Ent. Nemat., CABI, Wallingford, UK. p: 311-332.
Arthers, S., K.M. Heinz and J.R. Prasifka. 2004. An analysis of using entomopathogenic nematodes against above-ground pests. Bull. Entomol. Res. 94(4): 297-306.
Badii, K.B., M.K. Billah, N.K. Afreh, O.D. Obeng and G. Nyarko. 2015. Review of the pest status, economic impact and management of fruit infesting flies (Diptera:Tephritidae) in Africa. African J. Agric. Res. 10(12): 1488-1498.
Baig, M.B., F.S. Al-Subaiee and G.S. Straquadine. 2009. Role of Agricultural Extension in Sustainable Rural Development in Pakistan. Pub. By Banat University of Agricultural Sciences and Veterinary Medicine, Timisoara, Romania. Lucrari Stiintifice, Seria. 11(1): 291-308 pp.
Barbosa, C.R.C., M.S. Garcia, C. Dolinski, D. Bernardi and Nava D.E. 2009. Efficacy of indigenous entomopathogenic nematodes (Rhabditidae: Heterorhabditidae, Steinernematidae), from Rio Grande do Sul Brazil, against Anastrepha fraterculus Wied (Diptera: Tephritidae) in peach orchards. J. Invert. Patho. 102(1): 6-13
Beris, E.I., D.P. Papachristos, A. Fytrou. S.A. Antonnatos and D.C. Kontodimas. 2013. Pathogenicity of three Entomopathogenic fungi on pupac and adults of the Mediterranean fruit fly, Ceratitis capitata (Diptera: Tephritidae). J. Pest Sci. 86(2): 275-284.
Boiteau, G and C. Noronha. 2007. Topical, residual, and ovicidal contact toxicity of three reduced-risk insecticides against the European corn borer, Ostrinia nubilalis (Lepidoptera: Crambidae), on potato. Pest Manag. Sci. 63(12): 1230-1238.
Borges, R., M. Junior, R.M.I.C. Boff and M. Botton. 2015. Efeito de iscas tóxicas sobre Anastrepha fraterculus (Wiedemann) (Diptera: Tephritidae). Bio Assay. 10(1): 1-8.
Brown, A.E. 2006. Mode of action of insecticides and related pest control chemicals for production agriculture, ornamentalsa nd Maryland Cooperative Extension. p: 13.
Chen, P and H. Ye. 2007. Population dynamics of Bactrocera dorsalis (Diptera: Tephritidae) and analysis of factors influencing populations in Baoshanba, Yunnan, China. Entomol. Sci. 10(2): 141-147.
Devaraju, G., G.B. Narabenchi, C.R. Shruthi and A.M. Nadaf. 2018. Screening of ridge gourd genotypes aganist melon fruit fly, Bactrocera cucurbitae (Coquillett) under field conditions. J. Ent. Zoo. Stud. 6(4): 814-817.
Dhillon, M.K., J.S. Naresh, R. Singh and N.K. Sharma. 2005. The melon fruit fly, B. cucurbitae: A review of its biology and management. J. Insect Sci. 5(1): 40-60.
Dominiak, B.C. and P. Worsley. 2018. Review of cucumber fruit fly, Bactrocera cucumis (French) (Diptera: Tephritidae: Dacinae) in Australia: Part 1, host range, surveillance and distribution. Crop Protect. 106: 79-85.
Dominiak, B.C. 2018. Review of cucumber fruit fly, Bactrocera cucumis (French) (Diptera: Tephritidae: Dacinae): Part 2, biology, ecology and control in Australia. Crop Protect. 104: 35-40.
Deguine, J.-P., T. Atiama-Nurbel, J.-N. Aubertot, X. Augusseau, M. Atiama, M. Jacquot and B. Reynaud. 2015. Agroecological management of cucurbit-infesting fruit fly: a review. Agron. Sustain. Develop. 35(3): 937-965.
Galm, U and T.C. Sparks. 2015. Natural product derived insecticides: discovery and development of spinetoram. J. Indus. Micro. Biotech. 43(2-3): 185-193.
Guo W., X. Yan, G. Zhao and R. Han. 2017. Increased Efficacy of Entomopathogenic Nematode Insecticide Combinations against H. oblita (Coleoptera: Scarabaeidae). J. Eco. Entomol. 110(1): 41-51.
Hadapad, A.B., C.S. Prabhakar, S.C. Chandekar, J. Tripathi and R.S. Hire. 2016. Diversity of bacterial communities in the midgut of Bactrocera cucurbitae (Diptera: Tephritidae) populations and their potential use as attractants. Pest Manag. Sci. 72(6): 1222-1230.
Harter, W.D.R., A.D. Grutzmacher, D.E. Nava, R. Gonçalves and M. Botton. 2010. Isca tóxica e disrupção sexual no controle da mosca-das-frutas sul-americana e da mariposa-oriental em pessegueiro. Pes. Agro. Brasil. 45(3): 229-235.
Heve, W.K., E. Fahiem, El-Borai, D. Carrilloc and L. Duncan. 2017. Biological control potential of entomopathogenic nematodes for management of Caribbean fruit fly, Anastrepha suspensa Loew (Tephritidae). Pest. Mang. Sci. 73(6): 1220-1228.
Hossain, S. and M. Khalequzzaman. 2018. Repellent and oviposition deterrent activity of leaf extracts of Azadirachta indica A. Juss., Persicaria hydropiper (L.) Spach. and Vitex negundo Linn. against the melon fruit fly, Bactrocera cucurbitae (Coquillett) (Diptera: Tephritidae). J. Ent. Zoo. Stud. 6(2): 2291-2295.
Islam, S., D.M.S. Islam and R. Khanum. 2018. Economic impact of practicing IPM technology on Bitter Gourd (Momordica charantia L.) Production in comilla district of Bangladesh. Agri. 15(2): 11-23.
Jiang, Y and M. Feng. 2006. Compatibility of two fungal biocontrol agents’ conidia with commercial chemical acaricides. Chin. J. Appl. Ecol. 17(7): 1264-1268.
Joomaye, A.N., N.S. Price and J.M. Stonehouse. 2000. Quarantine pest risk analysis of fruit flies in Indian Ocean: the of Bactrocera zonata. Proceedings Indian Ocean Commission Regional Fruit Fly Symposium. p: 179-183.
Joseph, R.A., M.N. Borgia, M. Theresa and P. Miranda. 2017. Biocontrol potential of Heterorhabditis indica against the maggot of Bactrocera cucurbitae (Diptera:Tephritidae). Entomon. 42(1): 69-74.
Kamali, S.J., K.M. Hosseini, R. Campos-Herrera and L.W. Duncan. 2013. Biocontrol potential of the entomopathogenic nematodes Heterorhabditis bacteriophora and Steinernema carpocapsae on cucurbit fly, Dacus ciliatus (Diptera: Tephritidae). Bio. Sci. Techn. 23(11): 1307-1323.
Kate, A.O., R.K. Bharodi, M.D. Joshi, A.M. Pardeshi and R.R. Makadia. 2009. Seasonal incidence of fruit fly, Bactrocera cucurbitae (Coquillet) on cucumber. Asian Sci. 4(2): 83-84.
Kaya, H.K. and A.H. Hara. 1980. Differential susceptibility of Lepidopterous pupae to infection by the nematode Neoaplectana carpocapsae. J. Invert. Pathol. 36(3): 389-393.
Kaya, H.K. 1990. Soil ecology. In: Entomopathogenic nematodes in biological control. Gaugler R and Kaya HK (Ed.). CRC Press, Boca Raton, Florida. p: 93-115.
Khan, R.R., A.R Ali, A. Abid, M. Arshad, S. Majeed, S. Ahmed, A. Sajid Khan and M. Arshad. 2018. Compatibility of entomopathogenic nematodes (Nematoda: Rhabditida) and the biocide spinosad for mitigation of the armyworm, Spodoptera litura (F.) (Lepidoptera: Noctuidae) Egypt. J. Bio. Pest Cont. 28: 58.
Khursheed, S. and R. Desh. 2019. Response of fruit flies, Bactrocera spp. to different attractants in mid-hill Himalayas. J. Zool. Entomol. Stud. 7(1): 295-298.
Klungness, L.M., E.B. Jang, R.F.L. Mau, R.I. Vargas, J.S. Sugano and E. Fujitani. 2005. New sanitation techniques for controlling tephritid fruit flies (Diptera: Tephritidae) in Hawaii. J. Appl. Sci. Environ. Manage. 9(2): 5-14.
Koppenhofer, A.M and P.S. Grewal. 2005. Compatibility and interactions of EPN with other control agents. In P. S. Grewal, R. U. Ehlers, and D. I. Shapiro-Ilan (eds.), Nematodes as biocontrol agents. CABI, Wallingford United Kingdom. p: 363-381.
Koppenhofer, A.M., P.S. Grewal and H.K. Kaya. 2000. Synergism of imidacloprid and entomopathogenic nematodes against white grubs: the mechanism. Entomol. Exp. Appl. 94(3): 283- 293.
Lezama G.R., O.J. Molina, R.A. Pescador, V.E. Galindo, and R.E. Gonzalez. 2006. Efficacy of Steinernematid nematodes (Rhabditida: Steinernematidae) on the suppression of Anastrepha ludens (Diptera: Tephritidae) larvae in soil of differing textures: Laboratory and field trials. J. Agric. Urban Entomol. 23(1): 41-49.
Miller N.W., R.I. Vargas, R.J. Prokopy and B.E. Mackey. 2004. State-dependent attractiveness of protein bait and host fruit odor to Bactrocera cucurbitae (Diptera: Tephritidae) females. Ann. Entomol. Soc. Am. 97(5): 1063-1068.
Negrisoli, A.S., M.S. Garcia, C.R.C. Barbosa– Negrisoli, D. Bernardi and A. Silva. 2010. Efficacy of entomopathogenic nematodes (Nematoda: Rhabditida) and insecticide mixtures to control Spodoptera frugiperda (Smith, 1797) (Lepidoptera: Noctuidae) in corn crops. Crop Prot. 29(7): 677-683.
Pinero J.C., I. Jacome, R. Vargas and R.J. Prokopy. 2006. Response of female melon fly, Bactrocera cucurbitae, to host associated visual and olfactory stimuli. Entomol. Exp. Appl. 121(3): 261-269.
Poinar, J.R. 1990. Biology and taxonomy of Steinernematidae and Heterorhabditidae. In: Gaugler, R., Kaya, H. K. (Eds.) Ent. Nem.In Bio. Con., CRC Press, Boca Raton, FL. p: 23-62.
Quesada, M.E., P.V. Garcia and l.G. Jurado. 2012. The effect of temperature and soil moisture on the development of the pre-imaginal Mediterranean fruit fly (Diptera: Tephritidae). Environ. Entomol. 41(4): 966-970.
Rahman, M.S. and G.W. Norton.2019. Adoption and Impacts of Integrated Pest Management in Bangladesh: Evidence from Smallholder Bitter Gourd Growers. Hort. 5(2): 32-35.
Sapkota, R., K.C. Dahal and R.B. Thapa. 2013. Damage assessment and management of cucurbit fruit flies in spring-summer squash. J. Entomol. Nematol. 2(1): 7-12.
Shapiro D.I., D.H. Gouge, S.J. Piggott and J.P. Fife. 2012. Application technology and environmental considerations for use of entomopatogenic nematodes in biological control. Bio. Con. 38(1): 124-133.
Sirjani, F.O., Lewis, E.E and H.K Kaya. 2009. Evaluation of entomopathogenic nematodes against the olive fruit fly, Bacterocera oleae (Diptera: Tephritidae). Bio. Cont. 48(3): 23-36.
Yee, W.L. and L.A. Lacey. 2003. Stage-specific mortality of Rhagoletis indifferens (Diptera: Tephritidae) exposed to three species of Steinernema nematodes. Bio. Cont. 27(3): 349- 356.
Zimmerman, R.J and W.S. Cranshaw. 1990. Compatibility of three entomopathogenous nematodes (Rhabditida) in aqueous solutions of pesticides used in turf grass maintenance. J. Econ. Entomol. 83(1): 97-100.
Gazit, Y., Y. Rossler and I. Glazer. 2000. Evaluation of entomopathogenic nematodes for the control of mediterranean fruit fly (Diptera: Tephritidae). Biocontrol Sci. Technol. 10(2): 157-164.
Aatif, H.M., M.S. Hanif, M. Ferhan, M. Raheel, Q. Shakeel, W. Ashraf, M.I. Ullah and S. Ali. 2019. Assessment of the entomopathogenic nematodes against maggots and pupae of the oriental fruit fly, Bactrocera dorsalis (Hendel) (Diptera: Tephritidae), under laboratory conditions. Egyptian J. Biol. Pest Control. 29(1): 51.
Kapranas, A., I. Sbaiti, T. Degen and T.C.J. Turlings. 2020. Biological control of cabbage fly Delia radicum with entomopathogenic nematodes: Selecting the most effective nematode species and testing a novel application method. Biol. Control. 144: 104212.
Jaffuel, G., I. Sbaiti and T.C.J. Turlings. 2019. Encapsulated Entomopathogenic Nematodes Can Protect Maize Plants from Diabrotica balteata Larvae. Insects. 11(1): 27.
Means sharing different letters within each columns are significantly different at 5 % interval (P < 0.05, Tucky HSD test). Hi1‒ Heterorhabditis indica (100 IJs); Hi2‒ Heterorhabditis indica (150 IJs); SP: Spinosad; S1‒ Spinosad (30 mg); S2‒ Spinosad (40 mg).
According to Zimmerman and Cranshaw (1990) which reported high levels toxicity of chlorpyrifos to S. feltiae but with less toxicity to S. carpocapsae and heterorhabditid. Negrisoli et al. (2010) reported coaction between nematodes (S. glaseri and H. indica) with chlorpyrifos when targeting armyworm, S. frugiperda. According to Jiang and Feng (2006) long term compatibility between two agents (chlorpyrifos and the fungus), were observed as well as synergistic interaction between imidacloprid and white grubs. Koppenhofer et al. (2000) checked that the mechanisms laid to reduced pests activity because of the chemical, which allowed the nematode to overcome the insect’s defenses.
3.4. Effect of bio-control agents against developmental time of B. cucurbitae
For the first time the effect of nematodes and bacterial based spinosad was checked against developmental time of B. cucurbitae. When nematode H. indica and spinosad was applied in single doses the spinosad showed high infection rate which reduced the adult longevity, eggs laying capacity, eggs hatching percentage, fecundity rate, larval duration, pupal duration while nematodes also showed same results with little differences. Pupal survival, larval survival and pre-oviposition period was increased in spinosad treatments, which was observed low in nematodes.
In combine treatments spinosad and nematode with highest concentration (40g, 150IJs) showed gradually highest effect as compared to other treated combinations. While control treated with distilled water showed highest developmental rate. According to current results it was suggested that combination of nematodes and insecticides can provide a best control strategy as compared to others. This was first time study who reported these results, due to this it was becomes difficult to compare with other studies. However we want to compare our results with Guo et al. (2017); Khan et al. (2018) and Heve et al. (2017) who did some aspects of our current research and got significant results as compared to our study. According to reviews, and current research it is suggested that the best approaches to counteract with fruit flies is to use combined treatments of nematodes and bacterial based spinosad, which could provide effective results, and might be helpful at farmer level.
4. Conclusion
Present research was focused to control fruit flies species especially melon fruit fly with appropriate management tactics. In the past researchers largely considered pesticides applications for control of fruit flies, but unfortunately due to multiple application these pests has developed resistance against certain types of organophosphates. Broad spectrum applications of pesticides also imposed some global warming threats including human’s health issues. Under such circumstances current research was focused to control fruit flies species biologically rather than excessive pesticide application. Present research was based on use of EPNs with combination of bacterial based spinosad for successful control of fruit flies. Fruit flies larvae, pupae and adults was exposed to combine and single treatments of both currently applied bio-control agents. Effect of these both bio-control agents was also evaluated on the development of fruit flies. Obtained results suggested that both bio-control agents showed significant control of B. cucurbitae with expected mortalities. Bacterial based spinosad was observed more toxic than EPNs. In combined applications of both biocontrol agents significant results were obtained with causing maximum mortalities. As concentration was increased mortality rate was also increased. This research provides an environmentally safe fruit flies control technique. This research could provide a sound base for the effective management of fruit flies species.
List of Abbreviations: EPNs: Entomopathogenic nematodes, Hi: Heterorhabditis Indica, S: Spinosad, IPM: Integrated Pest Management, RH: Relative Humidity, IJs: Infective Juvenile stage, CRD: Complete Randomized Design, ANOVA: Analysis of Variance, SE: Standard Error.
Competing Interest Statement: This declare that all the authors of this research articles do not have any competing interest.
Author’s Contribution: All the author participate equally in this project. A.A. conducted experiments, while A.M. assisted for data analysis and experiments designs. S.S. assisted practically in experiments as well as in fruit flies collections from the orchards. M.I.M., S.S., C.L. and F. assisted to revise and improvements in the manuscript.
Acknowledgments: Author of this manuscript is thankful to all correspondence authors for help throughout the research during her Master study.
Funding Source: Research project was self-funded without any financial assistance.
References
Ahmad, G., A.A. Khan and S. Ansari. 2017. Interaction of a fly ash and root-knot nematode pathogens on Pumpkin (Cucurbita moschata Duch. ex Lam.). Tro. Plant. Res. 4(3): 449-455.
Akhurst, R and K. Smith. 2002. Regulation and safety. In: Gaugler, R. (Ed.), Ent. Nemat., CABI, Wallingford, UK. p: 311-332.
Arthers, S., K.M. Heinz and J.R. Prasifka. 2004. An analysis of using entomopathogenic nematodes against above-ground pests. Bull. Entomol. Res. 94(4): 297-306.
Badii, K.B., M.K. Billah, N.K. Afreh, O.D. Obeng and G. Nyarko. 2015. Review of the pest status, economic impact and management of fruit infesting flies (Diptera:Tephritidae) in Africa. African J. Agric. Res. 10(12): 1488-1498.
Baig, M.B., F.S. Al-Subaiee and G.S. Straquadine. 2009. Role of Agricultural Extension in Sustainable Rural Development in Pakistan. Pub. By Banat University of Agricultural Sciences and Veterinary Medicine, Timisoara, Romania. Lucrari Stiintifice, Seria. 11(1): 291-308 pp.
Barbosa, C.R.C., M.S. Garcia, C. Dolinski, D. Bernardi and Nava D.E. 2009. Efficacy of indigenous entomopathogenic nematodes (Rhabditidae: Heterorhabditidae, Steinernematidae), from Rio Grande do Sul Brazil, against Anastrepha fraterculus Wied (Diptera: Tephritidae) in peach orchards. J. Invert. Patho. 102(1): 6-13
Beris, E.I., D.P. Papachristos, A. Fytrou. S.A. Antonnatos and D.C. Kontodimas. 2013. Pathogenicity of three Entomopathogenic fungi on pupac and adults of the Mediterranean fruit fly, Ceratitis capitata (Diptera: Tephritidae). J. Pest Sci. 86(2): 275-284.
Boiteau, G and C. Noronha. 2007. Topical, residual, and ovicidal contact toxicity of three reduced-risk insecticides against the European corn borer, Ostrinia nubilalis (Lepidoptera: Crambidae), on potato. Pest Manag. Sci. 63(12): 1230-1238.
Borges, R., M. Junior, R.M.I.C. Boff and M. Botton. 2015. Efeito de iscas tóxicas sobre Anastrepha fraterculus (Wiedemann) (Diptera: Tephritidae). Bio Assay. 10(1): 1-8.
Brown, A.E. 2006. Mode of action of insecticides and related pest control chemicals for production agriculture, ornamentalsa nd Maryland Cooperative Extension. p: 13.
Chen, P and H. Ye. 2007. Population dynamics of Bactrocera dorsalis (Diptera: Tephritidae) and analysis of factors influencing populations in Baoshanba, Yunnan, China. Entomol. Sci. 10(2): 141-147.
Devaraju, G., G.B. Narabenchi, C.R. Shruthi and A.M. Nadaf. 2018. Screening of ridge gourd genotypes aganist melon fruit fly, Bactrocera cucurbitae (Coquillett) under field conditions. J. Ent. Zoo. Stud. 6(4): 814-817.
Dhillon, M.K., J.S. Naresh, R. Singh and N.K. Sharma. 2005. The melon fruit fly, B. cucurbitae: A review of its biology and management. J. Insect Sci. 5(1): 40-60.
Dominiak, B.C. and P. Worsley. 2018. Review of cucumber fruit fly, Bactrocera cucumis (French) (Diptera: Tephritidae: Dacinae) in Australia: Part 1, host range, surveillance and distribution. Crop Protect. 106: 79-85.
Dominiak, B.C. 2018. Review of cucumber fruit fly, Bactrocera cucumis (French) (Diptera: Tephritidae: Dacinae): Part 2, biology, ecology and control in Australia. Crop Protect. 104: 35-40.
Deguine, J.-P., T. Atiama-Nurbel, J.-N. Aubertot, X. Augusseau, M. Atiama, M. Jacquot and B. Reynaud. 2015. Agroecological management of cucurbit-infesting fruit fly: a review. Agron. Sustain. Develop. 35(3): 937-965.
Galm, U and T.C. Sparks. 2015. Natural product derived insecticides: discovery and development of spinetoram. J. Indus. Micro. Biotech. 43(2-3): 185-193.
Guo W., X. Yan, G. Zhao and R. Han. 2017. Increased Efficacy of Entomopathogenic Nematode Insecticide Combinations against H. oblita (Coleoptera: Scarabaeidae). J. Eco. Entomol. 110(1): 41-51.
Hadapad, A.B., C.S. Prabhakar, S.C. Chandekar, J. Tripathi and R.S. Hire. 2016. Diversity of bacterial communities in the midgut of Bactrocera cucurbitae (Diptera: Tephritidae) populations and their potential use as attractants. Pest Manag. Sci. 72(6): 1222-1230.
Harter, W.D.R., A.D. Grutzmacher, D.E. Nava, R. Gonçalves and M. Botton. 2010. Isca tóxica e disrupção sexual no controle da mosca-das-frutas sul-americana e da mariposa-oriental em pessegueiro. Pes. Agro. Brasil. 45(3): 229-235.
Heve, W.K., E. Fahiem, El-Borai, D. Carrilloc and L. Duncan. 2017. Biological control potential of entomopathogenic nematodes for management of Caribbean fruit fly, Anastrepha suspensa Loew (Tephritidae). Pest. Mang. Sci. 73(6): 1220-1228.
Hossain, S. and M. Khalequzzaman. 2018. Repellent and oviposition deterrent activity of leaf extracts of Azadirachta indica A. Juss., Persicaria hydropiper (L.) Spach. and Vitex negundo Linn. against the melon fruit fly, Bactrocera cucurbitae (Coquillett) (Diptera: Tephritidae). J. Ent. Zoo. Stud. 6(2): 2291-2295.
Islam, S., D.M.S. Islam and R. Khanum. 2018. Economic impact of practicing IPM technology on Bitter Gourd (Momordica charantia L.) Production in comilla district of Bangladesh. Agri. 15(2): 11-23.
Jiang, Y and M. Feng. 2006. Compatibility of two fungal biocontrol agents’ conidia with commercial chemical acaricides. Chin. J. Appl. Ecol. 17(7): 1264-1268.
Joomaye, A.N., N.S. Price and J.M. Stonehouse. 2000. Quarantine pest risk analysis of fruit flies in Indian Ocean: the of Bactrocera zonata. Proceedings Indian Ocean Commission Regional Fruit Fly Symposium. p: 179-183.
Joseph, R.A., M.N. Borgia, M. Theresa and P. Miranda. 2017. Biocontrol potential of Heterorhabditis indica against the maggot of Bactrocera cucurbitae (Diptera:Tephritidae). Entomon. 42(1): 69-74.
Kamali, S.J., K.M. Hosseini, R. Campos-Herrera and L.W. Duncan. 2013. Biocontrol potential of the entomopathogenic nematodes Heterorhabditis bacteriophora and Steinernema carpocapsae on cucurbit fly, Dacus ciliatus (Diptera: Tephritidae). Bio. Sci. Techn. 23(11): 1307-1323.
Kate, A.O., R.K. Bharodi, M.D. Joshi, A.M. Pardeshi and R.R. Makadia. 2009. Seasonal incidence of fruit fly, Bactrocera cucurbitae (Coquillet) on cucumber. Asian Sci. 4(2): 83-84.
Kaya, H.K. and A.H. Hara. 1980. Differential susceptibility of Lepidopterous pupae to infection by the nematode Neoaplectana carpocapsae. J. Invert. Pathol. 36(3): 389-393.
Kaya, H.K. 1990. Soil ecology. In: Entomopathogenic nematodes in biological control. Gaugler R and Kaya HK (Ed.). CRC Press, Boca Raton, Florida. p: 93-115.
Khan, R.R., A.R Ali, A. Abid, M. Arshad, S. Majeed, S. Ahmed, A. Sajid Khan and M. Arshad. 2018. Compatibility of entomopathogenic nematodes (Nematoda: Rhabditida) and the biocide spinosad for mitigation of the armyworm, Spodoptera litura (F.) (Lepidoptera: Noctuidae) Egypt. J. Bio. Pest Cont. 28: 58.
Khursheed, S. and R. Desh. 2019. Response of fruit flies, Bactrocera spp. to different attractants in mid-hill Himalayas. J. Zool. Entomol. Stud. 7(1): 295-298.
Klungness, L.M., E.B. Jang, R.F.L. Mau, R.I. Vargas, J.S. Sugano and E. Fujitani. 2005. New sanitation techniques for controlling tephritid fruit flies (Diptera: Tephritidae) in Hawaii. J. Appl. Sci. Environ. Manage. 9(2): 5-14.
Koppenhofer, A.M and P.S. Grewal. 2005. Compatibility and interactions of EPN with other control agents. In P. S. Grewal, R. U. Ehlers, and D. I. Shapiro-Ilan (eds.), Nematodes as biocontrol agents. CABI, Wallingford United Kingdom. p: 363-381.
Koppenhofer, A.M., P.S. Grewal and H.K. Kaya. 2000. Synergism of imidacloprid and entomopathogenic nematodes against white grubs: the mechanism. Entomol. Exp. Appl. 94(3): 283- 293.
Lezama G.R., O.J. Molina, R.A. Pescador, V.E. Galindo, and R.E. Gonzalez. 2006. Efficacy of Steinernematid nematodes (Rhabditida: Steinernematidae) on the suppression of Anastrepha ludens (Diptera: Tephritidae) larvae in soil of differing textures: Laboratory and field trials. J. Agric. Urban Entomol. 23(1): 41-49.
Miller N.W., R.I. Vargas, R.J. Prokopy and B.E. Mackey. 2004. State-dependent attractiveness of protein bait and host fruit odor to Bactrocera cucurbitae (Diptera: Tephritidae) females. Ann. Entomol. Soc. Am. 97(5): 1063-1068.
Negrisoli, A.S., M.S. Garcia, C.R.C. Barbosa– Negrisoli, D. Bernardi and A. Silva. 2010. Efficacy of entomopathogenic nematodes (Nematoda: Rhabditida) and insecticide mixtures to control Spodoptera frugiperda (Smith, 1797) (Lepidoptera: Noctuidae) in corn crops. Crop Prot. 29(7): 677-683.
Pinero J.C., I. Jacome, R. Vargas and R.J. Prokopy. 2006. Response of female melon fly, Bactrocera cucurbitae, to host associated visual and olfactory stimuli. Entomol. Exp. Appl. 121(3): 261-269.
Poinar, J.R. 1990. Biology and taxonomy of Steinernematidae and Heterorhabditidae. In: Gaugler, R., Kaya, H. K. (Eds.) Ent. Nem.In Bio. Con., CRC Press, Boca Raton, FL. p: 23-62.
Quesada, M.E., P.V. Garcia and l.G. Jurado. 2012. The effect of temperature and soil moisture on the development of the pre-imaginal Mediterranean fruit fly (Diptera: Tephritidae). Environ. Entomol. 41(4): 966-970.
Rahman, M.S. and G.W. Norton.2019. Adoption and Impacts of Integrated Pest Management in Bangladesh: Evidence from Smallholder Bitter Gourd Growers. Hort. 5(2): 32-35.
Sapkota, R., K.C. Dahal and R.B. Thapa. 2013. Damage assessment and management of cucurbit fruit flies in spring-summer squash. J. Entomol. Nematol. 2(1): 7-12.
Shapiro D.I., D.H. Gouge, S.J. Piggott and J.P. Fife. 2012. Application technology and environmental considerations for use of entomopatogenic nematodes in biological control. Bio. Con. 38(1): 124-133.
Sirjani, F.O., Lewis, E.E and H.K Kaya. 2009. Evaluation of entomopathogenic nematodes against the olive fruit fly, Bacterocera oleae (Diptera: Tephritidae). Bio. Cont. 48(3): 23-36.
Yee, W.L. and L.A. Lacey. 2003. Stage-specific mortality of Rhagoletis indifferens (Diptera: Tephritidae) exposed to three species of Steinernema nematodes. Bio. Cont. 27(3): 349- 356.
Zimmerman, R.J and W.S. Cranshaw. 1990. Compatibility of three entomopathogenous nematodes (Rhabditida) in aqueous solutions of pesticides used in turf grass maintenance. J. Econ. Entomol. 83(1): 97-100.
Gazit, Y., Y. Rossler and I. Glazer. 2000. Evaluation of entomopathogenic nematodes for the control of mediterranean fruit fly (Diptera: Tephritidae). Biocontrol Sci. Technol. 10(2): 157-164.
Aatif, H.M., M.S. Hanif, M. Ferhan, M. Raheel, Q. Shakeel, W. Ashraf, M.I. Ullah and S. Ali. 2019. Assessment of the entomopathogenic nematodes against maggots and pupae of the oriental fruit fly, Bactrocera dorsalis (Hendel) (Diptera: Tephritidae), under laboratory conditions. Egyptian J. Biol. Pest Control. 29(1): 51.
Kapranas, A., I. Sbaiti, T. Degen and T.C.J. Turlings. 2020. Biological control of cabbage fly Delia radicum with entomopathogenic nematodes: Selecting the most effective nematode species and testing a novel application method. Biol. Control. 144: 104212.
Jaffuel, G., I. Sbaiti and T.C.J. Turlings. 2019. Encapsulated Entomopathogenic Nematodes Can Protect Maize Plants from Diabrotica balteata Larvae. Insects. 11(1): 27.
Join Journal of Environmental and Agricultural Sciences (JEAS) Interested to join the JEAS Team Join JEAS as a member Editorial Board see Editors’ Responsibilities Join JEAS as a member Review Panel Reviewers’ Responsibilities (send your CV through email at editor.jeas@outlook.com)
JEAS Indexing Journal of Environmental EAS is indexed by reputed indexing services. Suggest Indexing service/s through email (editor.jeas@outlook.com)
Call for Articles Submit Your research for publication in the “Journal of Environmental and Agricultural Sciences (JEAS)” through email: editor.jeas@outlook.com
- How to prepare your manuscript before submission
- How to submit your manuscript
- Publication Ethics
- Publication Fee Currently JEAS is publishing manuscripts without publication or processing fee
JEAS Recently Published and Highly Cited Articles Citation record of JEAS: JEAS Google Scholar page Follow JEAS Facebook
