Journal of Environmental and Agricultural Sciences (JEAS). Jabeen et al., 2020. Volume 22(1): 64-70
Open Access – Research Article
Genotypic Variation in Micronutrient Composition of
Potato (Solanum tuberosum)
Beena Saeed 1,*, Ali Nawab 1, Sumayya Rani 2
1 Department of Agriculture (Agronomy), University, Sawabi, Sawabi, Pakistan
2 Department of Agriculture (Food Science and Technology), University, Sawabi, Sawabi, Pakistan
Abstract: To study micronutrient composition of different cultivars of potato, an experiment was conducted during 2015 at National Agriculture Research Centre, Islamabad, Pakistan. A set of five micronutrients [Copper (Cu), iron (Fe), Manganese (Mn), Boron (B), zinc (Zn)] in thirteen cultivars of potatoes collected from greenhouse of horticultural laboratory was used. Statistical analysis of the data revealed that maximum Cu concentration i.e. 8 mg kg-1 was estimated in cultivar 2 (Kennabee) whilst 4 mg kg-1 was the minimum Cu concentration in cultivars 1 (Caeser),11 (Cardinal),12 (SH-5),13 (Kuroda). The maximum Fe concentration 522 mg kg-1 was found in cultivar 9 (Ultimas) while minimum Fe concentration i.e. 296 mg kg-1 was observed in cultivar 6 (Agria). Similarly maximum Mn concentration 38mg kg-1 was estimated in cultivar 1 (Caeser) and the minimum Mn concentration i.e. 2 mg kg-1 in cultivar 2 (Kennabee). Likewise maximum Zn concentration 24 mg kg-1 was found in cultivar 11(Cardinal), while the minimum Zn concentration i.e. 6 mg kg-1 in cultivar 2(Kennabee). The maximum B concentration 3.68 mg kg-1 was estimated in cultivar 12 (SH-5) and the minimum B concentration i.e. 1.8 mg kg-1 was estimated in cultivars 7 (Sante) and 9 (Ultimas). Overall, all these results clearly showed significant differences of nutrient composition, i.e., Zn, Fe, Mn, Cu, and B among all the studied cultivars of potatoes.
Keywords: Cultivars of potato; chemical composition of potato; micronutrients.
*Corresponding author: Beena Saeed: drbeenasaeed@uoswabi.edu.pk
Cite this article as:
Saeed, B., A. Nawab and S. Rani. 2020. Genotypic variation in micronutrient composition of Potato (Solanum tuberosum). Journal of Environmental and Agricultural Sciences. 22(1):64-70. [Abstract] [View Full-Text] [Citations] 
Copyright © Saeed et al., 2020. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium provided the original author and source are appropriately cited and credited.
1. Introduction
Globally potato (Solanum tuberosum L.) is a major staple food crop (Haverkort and Struik, 2015; Stockem et al., 2020). For thousands of years potato remains major contributor of human diet and human health (Burgos et al., 2020). Potato tubers are of high nutritional value, as they are a good source of basic nutrients including carbohydrates, dietary fiber and vitamins (Burlingame et al., 2009). Moreover, potatoes are rich in minerals (boron, calcium, iron, magnesium, potassium, sodium and zinc) and phytochemicals (Furrer et al., 2018; Tian et al., 2016; Zaheer and Akhtar, 2016). In addition to its use as human food, potatoes are used for feed, industrial processing and seed. Among the industrial uses, mainly involved in alcohol and biofuel production, and making processed food products and starch (Hameed et al., 2018).
Diet and nutrition are essential in sustainable development goals (SDG) especially SDG3, good health and well being. In the recent know history potato remained an integral part of the global food system and it has the potential to ensure global food security and sustainable food energy in future (Devaux et al., 2020). Nutritional values of staple food play vital role in the emergence of dietetic, cancers and cardiovascular and other chronic diseases, which are one of the main reasons of premature mortalities across the world, especially in industrialized countries (Barclay et al., 2008; Herforth et al., 2019; Kelly et al., 2015).
In the recent years frequency of pathologic disorders has substantially increased, especially in less developed countries, and become a serious concern for public health (Zahoori, 2020). Moreover, contaminants and insufficient micronutrient intake reported to be the foremost cause of immature deaths and disabilities (Godecke et al., 2018). Hidden hunger describes condition of adequate intake of energy, however, malnutrition due to suboptimal consumption of micronutrients like iron, boron, zinc, and vitamin (Eggersdorfer et al., 2018; Sharma et al., 2017). Globally over 2 billion people are suffering from hidden hunger (Biesalski, 2013; Burchi et al., 2011).
Inadequate success in reducing chronic and hidden hunger, and related health issues are not sufficient to achieve targets of SDGs (Blesh et al., 2019; Godecke et al., 2018; Hendriks, 2018; Otekunrin et al., 2019). Hence, it will remain a global challenge to achieve SDGs with this limited success. Potato is rapidly replacing traditional staple foods (Camire et al., 2009; Mu et al., 2017; Zhang et al., 2017) therefore, it can be critical in achieving Sustainable Development Goals including SDG1 (No Poverty) SDG2 (Zero Hunger), SDG3 (Good Health and Well Being) and reduction of malnutrition through selection of cultivars with better yield and high mineral status or by mineral enrichment using biofortification (Rajiv and Kawar, 2016; Titcomb and Tanumihardjo, 2019; Yadava et al., 2018).
Assessment of genotypic nutritional variations can assist to improve nutritional status and biofortification of potato (Mu et al., 2017b; Rychcik et al., 2020; Vergara Carmona et al., 2019). This research was aimed to investigate genetic diversity among the potato cultivars for micronutrients (Zn, Fe, Mn, Cu, and B).
2. Materials and Methods
2.1. Plant Sampling
Thirteen cultivars of potatoes (1:Caeser, 2:Kennabee, 3:Satina, 4:Marfona, 5:Savalan, 6:Agria, 7:Sante, 8:Lala Faisal, 9:Ultimas, 10:Desire, 11:Cardinal, 12:SH-5, 13:Kuroda) were collected from greenhouse of Horticultural Research Institute, National Agricultural Research Centre (NARC), Islamabad and were brought to Land Resources Research Institute for processing and analysis. The plant samples were washed with tap water and then with distilled/deionized water. After washing, oven dried at 60oC. Samples were ground in a stainless steel Wiley mill and analyzed for nutrient content.
2.2. Plant Tissue Analysis
2.2.1. Wet digestion
For nutrients other than B, plant material was wet digested in a 2:1 mixture of nitric-perchloric acid (HNO3:HClO4). 0.25 g ground plant material was placed in 50 mL digestion flask. To this, 10 mL of acid mixture was added and the content of the flask was mixed by swirling, and left overnight.
The flask was placed on a hot plate in a digestion chamber and the temperature was gradually increased up to 230 0C. The flask was heated at this temperature until the production of brown HNO3 fumes ceases and dense white fumes of HClO4 appeared in the flask. The contents were further evaporated until the volume was reduced to about 3–5 mL, but not to dryness. The completion of digestion was confirmed when the liquid became colorless. Some distilled water was added after cooling the flask. Shift this liquid to 50 mL volumetric flask and make the volume. Filter this with the help of filter paper into storage bottles. Aliquots of this solution were used for the determination of P, K, Zn, Fe, Cu and Mn (Ryan et al,. 2001).
2.2.2. Zinc, iron, copper and manganese in digest
The determination of Zn, Fe, Cu, and Mn in plant digests was done by using atomic absorption spectroscopy (Wright et al., 1996).
2.2.3. Boron determination by dry ashing
Boron (B) in plant tissue was measured by dry ashing (Gaines and Mitchell, 1979) and subsequent measurement of B by colorimetry using azomethine-H (Keren, 1996). For dry ashing, 0.25 g dry, ground plant material was taken in a 30 mL porcelain crucible and placed in a muffle furnace. Plant material was ignited in furnace by slowly raising the temperature to 550°C. Ashing was continued for 6 hours after attaining 550°C. Ash was moistened with 5 drops of distilled water, and 10 mL 0.36 N sulfuric acid solution was added into the crucible. It was allowed to stand at room temperature for 1 hour, stirring occasionally with a plastic rod to break ash. Then the aliquot was filtered through whatman no. 42 filter paper and stored in plastic storage bottles. 1 mL of filtrate, 2 mL of buffer solution and 2 mL of color developing reagent (Azomethine-H and Lascorbic acid) was taken in Nalgene test tubes. The B concentration was measured on a Spectrophotometer at 420-nm using standard curve.
2.3. Statistical Analysis
The collected data was statistically analyzed using analysis of variance techniques appropriate for randomized complete block design. Means were compared using least significant difference (LSD) at 5% probability level (Jan et al., 2009).
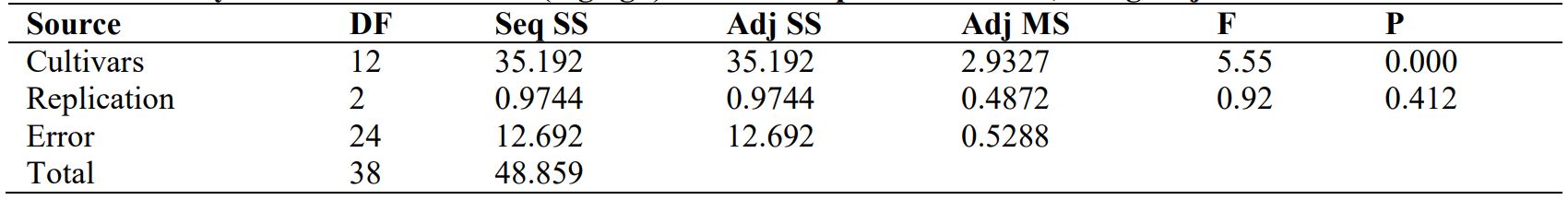
Table 1: Analysis of Variance for Cu (mg kg-1 ) in different potato cultivars, using Adjusted SS for Tests
3. Results
The Thirteen cultivars of potatoes were analyzed for Zn, Fe, Mn, Cu and B to determine nutrient composition and quality of various potato cultivars. Each individual nutrient has its specific role to enrich the edible quality of studied potato cultivars.

Fig. 1. Copper concentration in different potato cultivars
Copper
The significant analysis of variance for copper concentration in different cultivars of potatoes is illustrated in Table 1. The maximum Cu concentration i.e. 8 mg kg-1 was estimated in cultivar 2 whilst 4 mg kg-1 was the minimum Cu concentration in cultivars 1,11,12,13 (Fig. 1).
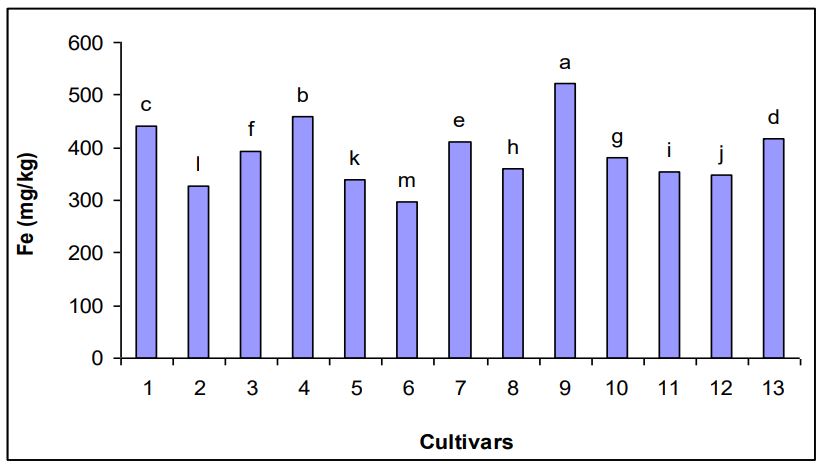
Fig. 2. Iron concentration in different potato cultivars
Iron
The significant analysis of variance for Fe concentration in different potato cultivars. The 522 mg kg-1 was the maximum Fe concentration found in cultivar 9 while minimum Fe concentration i.e. 296 mg kg-1 was observed in cultivar 6 (Fig. 2).
Manganese
The analysis of variance for Mn concentration in different cultivars of potatoes is presented in Table 3. The significant difference in Mn contents was observed in potato cultivars. The maximum Mn concentration 38 mg kg-1 was estimated in cultivar 1 and the minimum Mn concentration i.e. 2 mg kg-1was observed in cultivar 2 (Fig. 3).

Fig. 3. Manganese concentration in different potato cultivars
Zinc
Analysis of variance for Zn concentration in different cultivars of potatoes is illustrated in table 4. Significant difference was observed in Zn concentration. The maximum Zn concentration 24 mg kg-1 was found in cultivar 11 while the minimum Zn concentration i.e. 6 mg kg-1 in cultivar 2 (Fig. 4).

Fig. 4. Zn concentration in different potato cultivars.
Boron
The analysis of variance for boron concentration in different cultivars of potatoes is illustrated in Table 5. The significant difference in boron concentration was observed during the experiment. The maximum B concentration 3.68 mg kg-1 was estimated in cultivar 12 and the minimum boron concentration i.e. 1.8 mg kg-1 was estimated in cultivars 7 and 9 (Fig. 5).

Fig. 5. Born concentration in different potato cultivars.
4. Discussion
Plants require varying amounts of micronutrients during different stages of development. The availability of micronutrients depends on soil and environment. Zinc is relatively immobile nutrient and is concentrated in the soil organic matter near the soil surface. Cool wet weather reduces the availability of Zn, possibly resulting in a deficiency. The results found in this experiment had maximum Zn concentration (24 mg kg-1) in cultivar 11 (Cardinal), while the minimum Zn concentration i.e. 6 mg kg-1 in cultivar 2 (Kennabee) whose roots were weak and leaves were small in size than normal. These findings are similar with those of (Hamid and Asi, 2009) who, reported Zn deficiency in potato which is directly associated with root and shoot deformities, vascular system impairment and smaller leaf size.
Micronutrients availability generally decreases as the pH of soil increases. Availability of Zn, B, Cu declines rapidly as soil pH rises above 7. Similarly sandy soil shows more micronutrients deficiency than clay soils. Boron play vital role in sprouting, plant growth and tuber enlargement. The availability of B in soil is affected considerably by soil pH. At low pH, most of the B compounds are soluble but in case of sandy soil having low pH, B is lost down the profile by leaching if rainfall is high. Under drought condition the deficiency of B is observed due to low availability of B in sub-soils (Prasad et al,. 2014). Its deficiency causes the formation of a bushy plant with droopy leaves.
Manganese (Mn) and copper (Cu) are necessary for photosynthesis. The maximum Mn concentration 38 mg kg-1 was estimated in cultivar 1 (Caeser) and the minimum Mn concentration i.e. 2 mg kg-1 was observed in cultivar 2 (Kennabee). The reason might be that in cultivar 1 the tuber thickness and taste was substantial also the leaves were of healthier size. The main reason may be that the translocation of photosynthates from source to sink was maximum. These results are in agreement with those of (Prasad et al., 2014), who investigated that boron (B) aids in calcium translocation and helps maintain a balance between sugars and starch and also plays a role in cell wall strength, very similar to calcium (Ca). Similar reports were found that the application of sulfur and boron in deficient soil, increases the availability of boron in subsurface soil and promoted photosynthesis, leading to increased number of tubers per plant (Sharma, et al., 2011). In addition to these physiological roles, zinc, manganese and copper are keys to the potato crop’s phytoalexin (autoimmune) system for promoting disease tolerance.
Nutrient management is a critical component for successful potato production (Koch et al., 2020; Naumann et al., 2020). The use of micronutrients supplement should be based upon soil and tissues tests, and close visual examination of potato crop for deficiency symptoms (Ierna et al., 2017; Signh and Singh, 2019). These results are in agreement with those of (Robert and Tian, 2004), they concluded that the application of micronutrients through spray at different growth stages have commendable effect on performance of potato. Application of iron at various growth stages of potato enhance the vigor and viability of potato tubers as well as stem firmness. The maximum iron concentration in the research was found to be 522 mg kg-1 in cultivar 9 (Ultimas) while minimum Fe concentration i.e. 296 mg kg-1 was observed in cultivar 6 (Agria). The vigor of potato may be due to the transport of maximum iron concentration which directly enhanced mass of potato in cultivar 9. Likewise reports are presented by (Sud, 2006) who found that application of nitrogen, sulfur and iron can increase the mass and vigor of potato tuber as these nutrients enhanced the translocation and recovery of photosynthates.
Growers should carefully follow recommendations for micronutrients to avoid unnecessary costs and possible toxic effects on deleterious interactions with other nutrients. Selection of effective application method depends on micronutrients needed, local soil conditions and the stage in the growing season in which the deficiency is detected.
5. Conclusion
It can be concluded from the experiment that the approximation of the micronutrients boost up the chemical quality of potato as well as these micronutrients can enhance the vigor and yield of potato cultivars. Beside this, micronutrients are easily available in the local market; therefore the local farmers are recommended to bring these nutrients in use for general cultivation of potato.
List of Abbreviations: PSA – particle size analysis, PSD – particle size distribution, SOC: soil organic carbon; SOM: soil organic matter; CFI, clay flocculation index; FC: field capacity; PWP: permanent wilting point; SHC: saturated hydraulic conductivity.
Competing Interest Statement: The Authors declare that they have no competing interests regarding the contents of this paper.
Author’s Contribution: B.S. A.N. conceived the idea and designed the experiment. B.S., A.N. and S.R. wrote manuscript. All authors proofread and approved final draft of manuscript.
Acknowledgments: This study was conducted without any financial assistance. Assistance from colleagues at the University of Swabi to perform this study is highly appreciated.
References
Barclay, A.W., P. Petocz, J. McMillan-Price, V.M. Flood, T. Prvan, P. Mitchell and J.C. BrandMiller. 2008. Glycemic index, glycemic load, and chronic disease risk—A meta-analysis of observational studies. Am. J. Clin. Nutr. 87(3): 627-637.
Biesalski, H.K. 2013. Hidden Hunger. Springer Berlin Heidelberg, Berlin, Heidelberg, pp. 25-50.
Blesh, J., L. Hoey, A.D. Jones, H. Friedmann and I. Perfecto. 2019. Development pathways toward “zero hunger”. World Develop. 118: 1-14.
Burchi, F., J. Fanzo and E. Frison. 2011. The role of food and nutrition system approaches in tackling hidden hunger. Int. J. Env. Res. Public Health. 8(2): 358-373.
Burgos, G., T. Zum Felde, C. Andre and S. Kubow. 2020. The Potato and Its Contribution to the Human Diet and Health. In: Campos, H., Ortiz, O. (Eds.), The Potato Crop: Its Agricultural, Nutritional and Social Contribution to Humankind. Springer International Publishing, Cham, p. 37-74.
Burlingame, B., B. Mouillé and R. Charrondière. 2009. Nutrients, bioactive non-nutrients and antinutrients in potatoes. J. Food Comp. Anal. 22(6): 494-502.
Camire, M.E., S. Kubow and D.J. Donnelly. 2009. Potatoes and human health. Crit. Rev. Food Sci. Nutr. 49(10): 823-840.
Devaux, A., J.-P. Goffart, A. Petsakos, P. Kromann, M. Gatto, J. Okello, V. Suarez and G. Hareau. 2020. Global Food Security, Contributions from Sustainable Potato Agri-Food Systems. In: Campos, H., Ortiz, O. (Eds.), The Potato Crop: Its Agricultural, Nutritional and Social Contribution to Humankind. Springer International Publishing, Cham, pp. 3-35.
Eggersdorfer, M., U. Akobundu, R.L. Bailey, J. Shlisky, A.R. Beaudreault, G. Bergeron, R.B. Blancato, J.B. Blumberg, M.W. Bourassa, F. Gomes. 2018. Hidden hunger: Solutions for America’s aging populations. Nutrients. 10(9): 1210.
Furrer, A.N., M. Chegeni and M.G. Ferruzzi. 2018. Impact of potato processing on nutrients, phytochemicals, and human health. Crit. Rev. Food Sci. Nutr. 58(1): 146-168.
Gaines, T.P. and G.A. Mitchell. 1979. Boron determination in plant tissue by the azomethine H method. Common. Soil Sci. Plant Anal. 10: 1099– 1108.
Godecke, T., A.J. Stein and M. Qaim. 2018. The global burden of chronic and hidden hunger: Trends and determinants. Glob. Food Secur. 17: 21-29.
Hameed, A., S.S.-e.-A. Zaidi, S. Shakir and S. Mansoor. 2018. Applications of New Breeding Technologies for Potato Improvement. Front. Plant Sci. 9:925.
Hamid, A. and A.R. Asi. 1994. Effect of micronutrients on growth and yield of potato crop. Food Agric. Org. 8: 255-263.
Haverkort, A.J. and P.C. Struik. 2015. Yield levels of potato crops: Recent achievements and future prospects. Field Crops Res. 182: 76-85.
Hendriks, S.L. 2018. Food policy and nutrition economics in the SDG era. Agrekon. 57(3-4): 167-180.
Herforth, A., M. Arimond, C. Álvarez-Sánchez, J. Coates, K. Christianson and E. Muehlhoff. 2019. A Global Review of Food-Based Dietary Guidelines. Adv. Nutr 10(4): 590-605.
Ierna, A., A. Pellegrino and A. Malvuccio. 2017. Effects of micronutrient fertilization on the overall quality of raw and minimally processed potatoes. Postharvest Biol. Technol. 134: 38-44.
Jan, M.T., P. Roberts and D.L. Jones. 2009. Protein breakdown represents a major bottleneck in nitrogen cycling in grassland soils. Soil Bio. Biochem. 41(11): 2272-2282.
Kelly, J.T., D.P. Reidlinger, T.C. Hoffmann and K.L. Campbell. 2015. Telehealth methods to deliver multifactorial dietary interventions in adults with chronic disease: a systematic review protocol. Syst. Rev. 4(1): 185.
Keren, R. 1996. Boron. In: D.L. Sparks et al. (ed.) Methods of Soil Analysis, Part 3: Chemical Methods. Soil Science Society of America, Madison, WI, USA, p. 603–626.
Koch, M., M. Naumann, E. Pawelzik, A. Gransee and H. Thiel. 2020. The importance of nutrient management for potato production Part I: Plant nutrition and yield. Potato Res. 63(1): 97-119.
Mu, T., H. Sun, X. Liu. 2017a. Types of Potato Staple Food: A Brief Description. Potato Staple Food Processing Technology. Springer Singapore, Singapore, pp. 7-17.
Mu, T., H. Sun, X. Liu. 2017b. Improving the Nutritional Value of Potato Staple Foods. Potato Staple Food Processing Technology. Springer Singapore, Singapore, pp. 55-68.
Naumann, M., M. Koch, H. Thiel, A. Gransee and E. Pawelzik. 2020. The importance of nutrient management for potato production Part II: Plant nutrition and tuber quality. Potato Res. 63(1): 121- 137.
Otekunrin, O.A., O.A. Otekunrin, S. Momoh and I.A. Ayinde. 2019. How far has Africa gone in achieving the zero hunger target? Evidence from Nigeria. Glob. Food Secur. 22: 1-12.
Prasad, R., D. Kumar, Y.S. Shivay and D.S. Rana. 2014. Boron in Indian agriculture- a review. Indian J. Agron. 59(4): 511-517.
Rajiv, and P.G. Kawar. 2016. Enriched Potato for Mitigating Hidden Hunger. In: Singh, U., Praharaj, C.S., Singh, S.S., Singh, N.P. (Eds.), Biofortification of Food Crops. Springer India, New Delhi, pp. 433-457.
Robert, A. and W.U. Tian. 2004. Effect of foliar application of micronutrients on growth and performance of potato. Soil Sci. Soc. Am. 11: 1268- 1275.
Ryan, J., G. Estefan and A. Rashid. 2001. Soil and Plant Analysis Laboratory Manual. International Center for Agricultural Research in the Dry Areas (ICARDA), Aleppo, Syria. 172 pp.
Rychcik, B., J. Wierzbowska, M. KaźmierczakPietkiewicz, A. Światły. 2020. Impact of crop production system on the content of macronutrients in potato tubers. Acta Agric. Scand. B: Soil Plant Sci. https://doi.org/10.1080/09064710.2020.1734649.
Sharma, D.K., S.S. Khushwah and S.S. Rathore. 2011. Effect of sulphur on yield and quality of potato. Int. J. Agric.Res. 6(2): 143-148.
Sharma, P., P. Aggarwal and A. Kaur. 2017. Biofortification: A new approach to eradicate hidden hunger. Food Rev. Int. 33(1): 1-21.
Singh, P. and K. Singh. 2019. Role of micronutrients in potato cultivation. J. Pharmacognosy Phytochem. SP4:128-130.
Stockem, J., M. de Vries, E. van Nieuwenhuizen, P. Lindhout and P.C. Struik. 2020. Contribution and Stability of Yield Components of Diploid Hybrid Potato. Potato Res. https://doi.org/10.1007/s11540-019-09444-x
Sud, K..C. 2006. Anionic balance of phosphorus, sulphur and nitrogen in potato leaves under rainfed conditions in Shimla hills. J. Indian Soc. Soil Sci. 33(1): 68-73.
Tian, J., J. Chen, X. Ye and S. Chen. 2016. Health benefits of the potato affected by domestic cooking: A review. Food Chem. 202: 165-175.
Titcomb, T.J. and S.A. Tanumihardjo. 2019. Global Concerns with B Vitamin Statuses: Biofortification, Fortification, Hidden Hunger, Interactions, and Toxicity. Comprehensive Reviews in Food Science and Food Safety. 18(6): 1968-1984.
Vergara Carmona, V.M., A.B. Cecílio Filho, H.J.d. Almeida and P.L. Gratão. 2019. Fortification and bioavailability of zinc in potato. J. Sci. Food Agric. 99(7): 3525-3529.
Wright, R.J., and T.I. Stuczynski. 1996. Atomic absorption and flame emission spectrometry. Chemical Methods. Soil Sci. Soc.America, Madison.. 65–90.
Yadava, D.K., F. Hossain and T. Mohapatra. 2018. Nutritional security through crop biofortification in India: Status & future prospects. Indian J. Med. Res. 148(5): 621-631.
Zaheer, K. and M.H. Akhtar. 2016. Potato Production, Usage, and Nutrition—A Review. Crit. Rev. Food Sci. Nutr. 56(5): 711-721.
Zahoori, F.V. 2020. Nutrition and Diet. In: Zohoori FV, Duckworth RM (eds): The Impact of Nutrition and Diet on Oral Health. Monogr Oral Sci. Basel, Karger, 2020, vol 28, p 1–13.
Zhang, H., F. Xu, Y. Wu, H.-h. Hu and X.-f. Dai. 2017. Progress of potato staple food research and industry development in China. J. Integ. Agric. 16(12): 2924-2932.

Copyright © Saeed et al., 2020. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium provided the original author and source are appropriately cited and credited.
