Journal of Environmental and Agricultural Sciences (JEAS). Maqbool et al., 2016. Volume 9: 80-86
Open Access – Research Article
Inhibition of Cell Wall Degrading Enzymes and Improved Storability of Banana Fruit by Using Composite Edible Coatings
Mehdi Maqbool1,2,*, Noosheen Zahid1,2, Asgar Ali2, Ajit Singh2
1Department of Horticulture, Faculty of Agriculture, The University of Poonch, Rawalakot, Azad Jammu & Kashmir
2School of Biosciences, Faculty of Science, The University of Nottingham Malaysia Campus, Semenyih, 43500 Selangor, D.E., Malaysia
Abstract:
Composite effect of gum arabic (GA) (5, 10, 15 and 20%) plus 1.0% chitosan (CH) were investigated on cell wall degrading enzymes of banana stored at 13 ± 1 °C and 80 ± 3% relative humidity for 28 days and 5 days at simulated market conditions. Banana fruit coated with highest concentration of GA plus CH showed a significant (P<0.05) low concentration of cell wall degrading enzymes whereas the fruits coated with 5% GA plus 1.0% CH showed non-significant (P>0.05) difference with the control fruits. Fruits coated with 15 and 20% GA + 1.0% CH inhibited the production of cell wall degrading enzymes whilst an off flavour was recorded in these fruits. On the other hand fruits treated with 10% GA plus 1.0% CH showed a very gradual increase in cell wall degrading enzymes with a very uniform ripening of fruits. The findings of this study suggest that by using 10% GA plus 1.0% CH as an edible coating, the ripening process of banana can be delayed for up to 33 days while maintain the overall quality.
Keywords: Banana, Gum arabic, Chitosan, Enzymes, Post-harvest management
*Corresponding author: Mehdi Maqbool: mehdimaqbool@yahooo.com
Cite this article as: Maqbool, M., N. Zahid, A. Ali and A. Singh. 2016. Inhibition of cell wall degrading enzymes and improved storability of banana fruit by using composite edible coatings. Journal of Environmental & Agricultural Sciences. 9: 80-86. [View Full–Text] [Citations]. ResearchGate
Title: Inhibition of Cell Wall Degrading Enzymes and Improved Storability of Banana Fruit by Using Composite Edible Coatings Authors: Mehdi Maqbool, Noosheen Zahid, Asgar Ali, Ajit Singh Pages: 80-86
Copyright © Maqbool et al., 2016. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium provided the original author and source are appropriately cited and credited.
Inhibition of Cell Wall Degrading Enzymes, chitosan gum arabic banana coating, edible coating banana shelf life, polygalacturonase inhibition in banana, banana postharvest physiology
Introduction
Softening of fruit is considered an important aspect of quality which is noted during the process of ripening in various types of fruit (Ullah et al., 2015; Murayama et al., 2006). Perhaps it is caused by the collective effect of a range of modifications which occur in the network of polymers which make up the cell wall. These modifications take part in different ways to firmness loss and change in texture. Turgor pressure loss and cell wall degradation, contributes in firmness decrease in climacteric fruit (Zhou et al., 2008; Vogler et al., 2015; Romanazzi et al., 2016; Romanazzi et al., 2017). In most of the horticultural crops, the decrease in tissue firmness is considered an important quality parameter. The rate of fruit softening during the process of ripening may affect the quality of fruit as well as its life in storage (Ali et al., 2004). Fruit cell membrane integrity, which keeps the turgor pressure in fruit, can be destroyed by reactive oxygen species (ROS) which are produced in storage (Song et al., 2016). It is a fact that with ageing the anti-oxidative activity in fruit slows down, however, with an efficient antioxidant system the process of senescence can be delayed (Zheng et al., 2007; Sakif et al., 2016). Moreover, it has also been ascribed that the degradation of cell wall composition and structure disassembly happens due to the action of certain enzymes (among other things), which include polygalacturonase (PG), pectin methylesterase (PME) and cellulase on polysaccharides, such as pectin, hemicelluloses and cellulose, present in the cell wall (Cheng et al., 2009; Fraeye et al., 2007). However, their mode of action varies noticeably with fruit type (Steinhauser et al., 2010; Kojima et al., 1994). In case of banana, it is very little known about the role and importance of these fruit softening enzymes.
Edible coatings have been very well studies and are known for their capabilities to retard the softening process as well as metabolic activities (Hewajulige et al., 2015; Ali et al., 2011; Conforti and Zink, 2002; Park, 1999; Xing et al., 2016). Chitosan (CH) has been widely used in plant systems (Malerba and Cerana, 2016; Vieira et al., 2016; Xing et al., 2016), and also as an edible coating for fresh fruits and vegetables to retain their texture (Ali et al., 2011; Romanazzi et al., 2002; Kerch, 2015; Versino et al., 2016). However, there is limited information available on the use of gum arabic (GA) as an edible coating on fresh fruits and vegetables. Our group has tested this natural compound on fresh produce in recent years and this study is the part of that big project.
In the present study, a combination of both GA and CH was selected to investigate the effect of edible composite coatings on cell wall degrading enzymes such as polygalacturonase (PG), pectin methylesterase (PME) and β-galactosidase (β-gal) in banana fruit during storage, with reference to fruit firmness results published previously by our group (Maqbool et al., 2011).
2. Materials and Methods
Banana fruit (AAA group, cv. Pisang Berangan) were collected from a local orchard located at Beranang, Semenyih, Malaysia. Uniform fruits of equal size, colour and no disease symptoms were selected and transported to laboratory within 1 hour. All the solutions of GA [5, 10, 15, 20% (w/v)] incorporated with 1.0% CH were prepared as described by Maqbool et al. (2011). Commercially mature green banana fruit were dipped in these coating solutions for 2-3 min, to ensure the uniform application of coating solutions on the whole surface. The excess coating solution on the fruit was allowed to drain off. Fruit were then air dried at room temperature (25 ± 3°C) with the aid of a fan and kept in corrugated cardboard boxes. Boxes were randomly stored in a cold room set at 13 ± 1°C with 85 ± 5% RH. Observations were recorded at intervals of 7 days for 28 days and then the fruit were transferred to room temperature (25 ± 3°C) for 5 days.
2.1 Determination of fruit firmness and activity of cell wall degrading enzymes
2.1.1 Crude enzyme extraction
The method of Lazan et al. (1989) was used to prepare a crude enzyme. Fruit samples (10 g pulp) from each treatment were mashed at 4°C using a mortar and pestle in 10 ml of 0.1 M sodium citrate buffer (pH 4.6), containing 1 M NaCl, 13 mM EDTA, 10 mM β-mercaptoethanol and 1% (w/v) PVP-40. Sample mixture was homogenised for 1 h on ice and then centrifuged at 12,000 × g for 30 min at 4°C. Volume of supernatant was measured and then used as a crude enzyme extract for determining the activity of PG, PME and β-gal enzymes.
2.1.2 Assay of polygalacturonase (PG) activity
PG activity was measured by the release of reducing sugars using 2-cyanoacetamide as described by Gross (1982) with some modifications. The reaction was started by adding 0.75 ml of 1.5% (w/v) polygalacturonic acid (pH 5.2) and 0.1 ml of 0.6 M NaCl and the sample mixture was placed in a water bath for 10 min at 37°C. Then 0.15 ml of filtered extract (supernatant) was added to the reaction mixture and again placed in the water bath at 37°C for 60 min. The reaction was stopped by adding 2.5 ml of 100 mM sodium borate (pH 9.0) and 0.5 ml of 1% (w/v) 2-cyanoacetamide and then boiled for 10 min, followed by immediate incubation on ice. The content of reducing sugar produced was determined using a spectrophotometer (Model: Biochrom Libra S12; Biochrom Ltd., Cambridge, UK) at 276 nm. For a control, the crude extract was boiled for 10 min. A standard curve was prepared using 0.5 mM α-D-monogalacturonic acid stock solution. The enzyme activity was expressed as nmol of galacturonic acid released per second (expressed as nkatal g-1 FW). The amount of enzyme required to liberate 1 nmol of galacturonic acid per second per gram of fresh fruit weight is termed as one unit.
2.1.3 Assay of pectin methylsterase (PME) activity
Assay of PME was carried out according to the method described by Awad and Young (1979). Crude extract (0.5 ml) was added to 25 ml of 1% (w/v) pectin (pH 7.3) solution that contained 0.3M NaCl. The reaction mixture was titrated against 0.01M NaOH until the pH reached 7.3 and maintained for 10 min. pH was measured using a glass electrode pH meter (Model: CyberScan pH 510, Eutech Instruments Pte Ltd, Singapore). After 10 min, the volume of NaOH which maintained a pH of 7.3 was measured. Crude extract boiled for 10 min was used as a control. Enzyme activity was expressed as nano-equivalents of ester hydrolysed per second per gram of fresh fruit weight (nequiv. s-1 g-1 FW).
2.1.4 Assay of β-galactosidase (β-gal) activity
Activity of β-gal enzyme was measured according to the method given by Pressey (1983). 0.4 ml of 0.1% (w/v) bovine serum albumin and 0.4 ml of 13 mM substrate p-nitrophenyl-β-D-galactopyranoside were added to 0.52 ml 0.1 M tri-sodium citrate buffer (pH 4.0). Mixture was pre-incubated at 37°C for 10 min. Then 80 µl of crude enzyme extract was added to the mixture and incubation was continued in the water bath for 15 min at 37°C. The reaction was stopped by adding 2 ml of 0.2 M sodium carbonate. The reaction mixture was mixed thoroughly and the p-nitrophenol content produced was determined using a spectrophotometer (Model: Biochrom Libra S12; Biochrom Ltd., Cambridge, UK) at 415 nm. Crude extract boiled for 10 min was used as a control during this assay. Standard curve was prepared using 0.5 mM p-nitrophenol stock solution. Activity of enzyme was expressed as nkatal p-nitrophenol (nmol per second) per gram fresh fruit weight (nkatal g-1 FW).
2.2 Statistical analysis
Experiment was arranged in a completely randomized design (CRD) with four replicates, each consisting of 20 fruit. Data was analyzed using analysis of variance (ANOVA) and significant differences among treatments were tested by the least significant difference (LSD) test at (P≤0.05) using MSTAT-C software. Entire experiment was repeated twice, in order to confirm the reproducibility of results.
3. Results
The increase in activity for all the cell wall degrading enzymes tested was significantly (P≤0.05) delayed in fruit coated with GA plus CH as compared to the control fruit during and after storage.
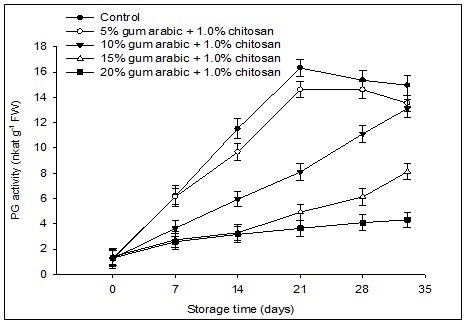
Fig 1. Effect of various concentrations of gum arabic incorporated with chitosan on polygalacturonase (PG) activity of banana fruit during storage (13°C, 80% RH) for 28 days followed by 5 days at simulated marketing conditions (25°C, 60% RH). The vertical bars represent the standard error of means for four replicates.
A general increase in PG activity was recorded in all the treatments (Fig 1). A highest increase in PG activity was recorded in control fruit and the fruit treated with 5% GA plus 1.0% CH on day 21st and slightly decreased afterwards. Whereas, fruit treated with 10% GA plus 1.0% CH showed the continuous increase in the activity of PG even up to 33 days of storage. In contrast, a very small but constant increase in PG activity was observed in 15 and 20% GA plus 1.0% CH coated fruit. A negative correlation was observed between PG activity and fruit firmness (r = -0.88; P≤0.05) during storage. From r2 value {(0.88)2 x 100}, it is clear that every unit increase in PG activity results in 77.4% decrease in firmness of banana fruit.
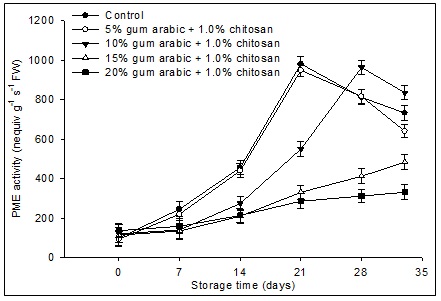
Fig 2. Effect of various concentrations of gum arabic incorporated with chitosan on pectin methylesterase (PME) activity of banana fruit during storage (13°C, 80% RH) for 28 days followed by 5 days at simulated marketing conditions (25°C, 60% RH). The vertical bars represent the standard error of means for four replicates.
A significant (P<0.05) maximum increase in PME activity was recorded in control fruit and the fruit treated 5% GA plus 1.0% CH on day 21, while in fruit treated with 10% GA plus 1.0% CH the highest activity was observed on day 28, after which there was a slight decrease in all the three treatments (Fig. 2). In contrast, PME activity increased at a slower rate in 15 and 20% GA plus 1.0% CH coated fruit during the complete storage period. The PME activity in banana fruit was negatively correlated with fruit firmness (r = -0.79, P≤0.05) during storage. From r2 value {(0.79)2 x 100} the increase in PME activity reduced the fruit firmness up to 62.4%.
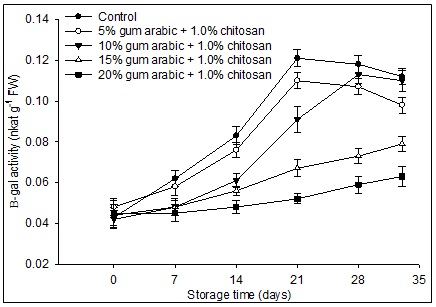
Fig 3. Effect of various concentrations of gum arabic incorporated with chitosan on β-galactosidase (β-gal) activity of banana fruit during storage (13°C, 80% RH) for 28 days followed by 5 days at simulated marketing conditions (25°C, 60% RH). The vertical bars represent the standard error of means for four replicates.
β-gal activity in control and 5% GA plus 1.0% CH coated fruit increased and reached its maximum on day 21 while in the fruit treated with 10% GA plus 1.0% CH the maximum activity was observed on day 28 but after that there was a slight decrease in all the three treatments (Fig. 3). A similar pattern of β-gal activity was recorded in the fruit treated with 15 and 20% GA plus 1.0% CH. β-gal activity is negatively correlated with fruit firmness (r = -0.854, P≤0.05) during storage. From r2 value {(0.854)2 x 100} contribution of β-gal activity to variation in firmness is 72.9%.
.
4. Discussion
Fruit firmness is considered an important parameter as it affects the postharvest quality and shelf-life of fruit. In a previous study by our group it was found that softening was greatly reduced by more concentrated GA with CH coatings (Maqbool et al., 2011). Control and 5% GA with 1.0% CH treated fruit lost their textural integrity and suffered collapse of tissues earlier than the fruit treated with higher concentrations.
Actually, the softening of fruit happens due to the collapse of cell structure and deterioration of cell wall composition (Ali et al., 2013). It is a bio-chemical process which happens due to the involvement of certain enzymes which cause the hydrolysis of pectin and starch. These enzymes include PG, PME and β-gal, which can play an important role in the softening of fruit (Lazan et al., 1995). This turgor loss in fruits is a physical function regulated by the physical barrier (covering) over the fruit (Yeats and Rose, 2013). In our results, the maintenance of fruit firmness at higher GA concentration is due to the fact that the coating helped in maintaining turgor pressure and reduced the cell wall deterioration.
To investigate the role of GA and CH in maintaining fruit firmness some enzymes activities were studied during this experiment. As fruit softening involves the cell wall hydrolases such as solubility of pectin which occurs with increase in PG activities (Yaman and Bayoindirli, 2002). The primary softening of banana is characterized by enhanced solubilization of cell wall pectin (Lazan et al., 1995).
The activity of PG in this study was found to be very low in fruit treated with the higher concentrations of GA with CH as compared to the control and 5% GA plus 1.0% CH. Whereas, at 10% GA + 1.0% CH gives a very gradual increase in PG activity which might be due to the reason that this combination coated the fruit cuticle in a very precise manner and helps to maintain its firmness (Maqbool et al., 2011).
Results suggests that the composite coatings (GA + CH) are helpful in reducing the PG activity and the similar results were shown by Ruoyi et al. (2005) on chitosan coated peach fruit. As chitosan is a negatively charged polysaccharide and directly adheres to positively charged pectin and forms chitosan-pectin. This chitosan-pectin has the ability to prevent the access of PG to cell wall substrate and help in maintenance of fruit firmness (Gol et al., 2013).
PME is an important enzyme which plays a critical role in softening of fruit tissues (Prasanna et al., 2007). During fruit ripening, it is found to be responsible for de-esterification of the highly methyl-esterified polygalacturonans in the cell wall (Payasi et al., 2009). PME is also responsible for the hydrolyzation of pectins to demethylated pectins which are more easily solubilized PG and results in pectin degradation (Zhou et al., 2011).
In our results the control fruit and fruit coated with 5% GA + 1.0% CH showed a higher activity of PME on day 21 in comparison with the fruits coated with higher concentration of composite coatings. This lower activity of PME in coated fruits might be responsible for the brittleness of fruit during storage (Goh et al., 2013; Gonzalez-Aguilar et al., 2009). It has been reported that edible coatings has the ability to reduce several enzymes which are responsible for cell wall degradation (Bhaskar-Reddy et al., 2000).
β-gal is also important enzyme which plays a significant role in firmness loss and cell wall modification as it is allied with the solubility of glycan matrix and also pectin degrading enzymes (Harpster et al., 2002; Tieman et al., 1992). In this study the continuous increase in β-gal activity in untreated fruit highlighted the fact that uncoated fruit tend to lose their firmness more than the treated fruit. These results are in line with Gonzalez-Aguilar et al. (2009) and Gol et al. (2013) who showed that edible coatings provide a mechanical strength to the fruit by inhibiting the enzyme activity and this inhibition in enzymes activity could be interconnected with the physiological changes induced by the treatments.
In our results, higher concentrations of GA and CH coatings retained the maximum firmness and fruit remained green until day 33 of the storage which might be due to the reason that at higher concentrations the viscosity of coating solutions was very high (Maqbool et al., 2011). These highly viscous solutions formed a very heavy layer on fruit surface which may blocked the respiration process resulting in reduced ripening process but off-flavour was noted in these fruits. Similar results were recorded by Ali et al. (2010) when they treated tomato fruit with GA solutions. El-Anany et al. (2009) also found the similar results on ‘Anna’ apples with GA based coatings.
Conclusion
In conclusion, softening increased with storage; especially in the control and 5% GA with 1.0% CH coated fruit. The maintenance of firmness in banana fruit coated with 10, 15 and 20% GA plus 1.0% CH composite coatings could be due to the slower enzymatic activity of cell wall degrading enzymes and thus suppressing the respiration process and other ripening related activities during storage. In addition, 10% GA + 1.0% CH is the best combination to slow down the enzymatic activity without any off-flavour. Hence, the combination of these coatings might be useful in future for improving postharvest quality and shelf-life of banana fruit.
Key Findings:
- 10% GA + 1% chitosan = optimal
- Shelf life extended up to ~33 days
- Higher concentrations → off-flavor
Acknowledgements: The authors are thankful to the Ministry of Agriculture (MOA) Malaysia for provision of funds to support this project under grant no. (05-02-12-SF0031) and also Jumbo Trading Company, based at Thailand as they supplied GA for these trials.
Competing Interests: The authors declare that there is no potential conflict of interest.
Inhibition of Cell Wall Degrading Enzymes, chitosan gum arabic banana coating, edible coating banana shelf life, polygalacturonase inhibition in banana, banana postharvest physiology
References
Ali, A., N. Zahid, S. Manickam, Y. Siddiqui, and P.G. Alderson. 2013. Double layer coatings: A new technique for maintaining physico-chemical characteristics and antioxidant properties of dragon fruit during storage. Food Bioprocess Technol. 7: 2366-2374.
Ali, A., T.M.M. Mahmud, S. Kamaruzaman and Y. Siddiqui. 2011. Effect of chitosan coatings on the physicochemical characteristics of Eksotika II papaya (Carica papaya L.) fruit during cold storage. Food Chem. 124: 620-626.
Ali, Z.M., L.H. Chin and H. Lazan. 2004. A comparative study on wall degrading enzymes, pectin modifications and softening during ripening of selected tropical fruits. Plant Sci. 167: 317-327.
Awad, M. and R.E. Young. 1979. Postharvest variation in cellulase, polygalacturonase, and pectinmethylesterase in avocado (Persea americana Mill, cv. Fuerte) fruits in relation to respiration and ethylene production. Plant Physiol. 64: 306-308.
Bhaskar-Reddy, M.V., P. Angers, F. Castaigne and J. Arul. 2000. Chitosan effects on black mold rot and pathogenic factors produced by Alternaria alternate in postharvest tomatoes. J. Am. Soc. Hort. Sci. 125: 742-747.
Cheng, G., X. Duan, Y. Jiang, J. Sun, S. Yang and B. Yang. 2009. Modification of hemicellulose polysaccharides during ripening of postharvest banana fruit. Food Chem. 1151: 43-47.
Conforti, F.D. and J.B. Zinck. 2002. Hydrocolloid-lipid coating effect on weight loss, pectin content, and textural quality of green bell peppers. J. Food Sci. 67: 1360-1363.
Fraeye, I., A.D. Roeck, T. Duvetter, I. Verlent, M. Hendrick and A.V. Loey. 2007. Influence of pectin properties and processing conditions on thermal pectin degradation. Food Chem. 105: 555-563.
Gol, N.B., R. Pooja, T.V. Patel and R. Rao. 2013. Improvement of quality and shelf-life of strawberries with edible coatings enriched with chitosan. Postharvest Biol. Technol. 85: 185-195.
Gonzalez-Aguilar, G.A., E. Valenzuela-Soto, J. Lizardi-Mendoza, F. Goycoolea, M.A. Martinez-Tellez, M.A. Villegas-Ochoa, I. Monroy-Garcia and J.F. Ayala-Zavala. 2009. Effect of chitosan coating in preventing deterioration and preserving the quality of fresh-cut papaya ‘Maradol’. J. Sci. Food Agric. 89: 15-23.
Gross, K.C. 1982. A rapid and sensitive spectrophotometric method for assaying polygalacturonase using 2-cyano-acetamide. Hort. Sci. 17: 933-934.
Harpster, M.H., D.A. Brummell and P. Dunsmuir. 2002. Suppression of ripening-related endo-1,4-β-glucanase in transgenic pepper fruit does not prevent depolymerization of cell wall polysaccharides during ripening. Plant Mol. Biol. 50: 345-355.
Hewajulige, I.G.N., R.S. Wilson Wijeratnam, M.G.D.S., Perera and S.A. Fernando. 2015. Extending storage life of commercially important tropical fruits using bio-waxes. Acta Horticul. 1091: 283-290.
Kerch, G. 2015. Chitosan films and coatings prevent losses of fresh fruit nutritional quality: A review. Trends Food Sci. Technol. 46(2-A): 159-166.
Kojima, K., N. Sakurai and S. Kuraishi. 1994. Fruit softening in banana: correlation among stress-relaxation parameters, cell wall components and starch during ripening. Physiol. Planta. 90: 772-778.
Lazan, H., M.K. Selamat and Z.M. Ali. 1995. β-Galactosidase, polygalacturonase and pectinesterase in differential softening and cell wall modification during papaya fruit ripening. Physiol. Planta. 95: 106-112.
Lazan, H., Z.M. Ali, K.S. Liang and K.L. Yee. 1989. Polygalacturonase activity and variation in ripening of papaya fruit with tissue depth and heat treatment. Physiol. Planta. 77: 93-98.
Malerba, M. and R. Cerana. 2016. Chitosan effects on plant systems. Int. J. Mol. Sci. 17: 996.
Maqbool, M., A. Ali, P.G. Alderson, N. Zahid and Siddiqui, Y. 2011. Effect of a novel edible composite coating based on gum arabic and chitosan on biochemical and physiological responses of banana fruits during cold storage. J. Agric. Food Chem. 59: 5474-5482.
Murayama, H., T. Katsumata, H. Endou, T. Fukushima and N. Sakurai. 2006. Effect of storage period on the molecular-mass distribution profile of pectic and hemicellulosic polysaccharides in pears. Postharvest Biol. Technol. 40: 141-148.
Payasi, A., N.N. Mishra and A.L. Soares-Chaves. 2009. Biochemistry of fruit softening: An overview. Physiol. Mol. Biol. Plants. 15: 103-113.
Prasanna, V., T.N. Prabhaand and R.N. Tharanathan. 2007. Fruit ripening phenomena-an overview. Critic. Rev. Food Sci. Nutr. 47: 1-19.
Pressey, R. 1983. β-Galactosidases in ripening tomatoes. Plant Physiol. 71: 132-135.
Romanazzi, G., E. Feliziani, S.B. Baños and D. Sivakumar. 2017. Shelf life extension of fresh fruit and vegetables by chitosan treatment. Crit. Rev. Food Sci. Nutr. 57(3): 579-601.
Romanazzi, G., F. Nigro, A. Ippolito, D. Di Venere and M. Salerno. 2002. Effects of pre- and postharvest chitosan treatments to control storage grey mold of table grapes. J. Food Sci. 67: 1862-1867.
Romanazzi, G., S.M. Sanzani, Y. Bi, S. Tian, P.G. Martínez and N. Alkan. 2016. Induced resistance to control postharvest decay of fruit and vegetables. Postharvest Biol. Technol. 122: 82-94.
Ruoyi, K., Y. Zhifang and L. Zhaoxin. 2005. Effect of coating and intermittent warming on enzymes, soluble pectin substances and ascorbic acid of Prunus persica (Cv. Zhonghuashoutao) during refrigerated storage. Food Res. Int. 38: 331-336.
Sakif, T.I., A. Dobriansky, K. Russell and T. Islam. 2016. Does chitosan extend the shelf life of fruits? Adv. Biosci. Biotechnol. 7(8): 6.
Song, H., W. Yuan, P. Jin, W. Wang, X. Wang, L. Yang and Y. Zhang. 2016. Effects of chitosan/nano-silica on postharvest quality and antioxidant capacity of loquat fruit during cold storage. Postharvest Biol. Technol. 119: 41-48.
Steinhauser, M.C., D. Steinhauser, K. Koehl, F. Carrari, Y. Gibson, A.R. Fernie and M. Stitt. 2010. Enzyme activity profiles during fruit development in tomato cultivars and Solanum pennellii. Plant Physiol. 153: 80-98.
Tieman, D.M., R.W. Harriman, G. Ramamohan and A.K. Handa. 1991. An antisense pectin methylesterase gene alters pectin chemistry and soluble solids in tomato fruit. Plant Cell. 4: 667-679.
Ullah, S., A.S. Khan, A.U. Malik, M. Shahid and K. Razzaq. 2015. Cultivar, harvest location and cold storage influence fruit softening and antioxidative activities of peach fruit [Prunus persica (L.) Batsch.]. Pak. J. Bot. 47: 699-709.
Versino, F., O.V. Lopez, M.A. Garcia and N.E. Zaritzky. 2016. Starch-based films and food coatings: An overview. Starch – Stärke. 68(11-12): 1026-1037.
Vieira, J.M., M.L. Flores-López, D.J. de Rodríguez, M.C. Sousa, A.A. Vicente and J.T. Martins. 2016. Effect of chitosan–Aloe vera coating on postharvest quality of blueberry (Vaccinium corymbosum) fruit. Postharvest Biol. Technol. 116: 88-97.
Vogler, H., D. Felekis, B.J. Nelson and U. Grossniklaus. 2015. Measuring the mechanical proeprties of plant cell walls. Plants. 4: 167-182.
Xing, Y., Q. Xu, X. Li, C. Chen, L. Ma, S. Li, Z. Che and H. Lin. 2016. Chitosan-based coating with antimicrobial agents: preparation, property, mechanism, and application effectiveness on fruits and vegetables. Int. J. Polym. Sci. 2016: 24.
Yaman, Ö. and L. Bayoindirli. 2002. Effects of edible coating and cold storage on shelf-life and quality of cherries. Lebensmittel-Wissenschaft und-Technologie. 35: 146-150.
Yeats, T.H. and J.K.C. Rose. 2013. The formation and function of plant cuticles. Plant Physiol. 163: 5-20.
Zheng, X., S. Tian, X. Meng and B. Li. 2007. Physiological and biochemical responses in peach fruit to oxalic acid treatment during storage at room temperature. Food Chem. 104: 156-162.
Zhou, R., Y. Li, L. Yan and J. Xie. 2011. Effect of edible coatings on enzymes, cell membrane integrity, and cell-wall constituents in relation to brittleness and firmness of Huanghua pears (Pyrus pyrifolia Nakai, cv. Huanghua) during storage. Food Chem. 124: 569-575.
Zhou, R., Y. Mo, Y. Li, Y. Zhao, G. Zhang and Y. Hu. 2008. Quality and internal characteristics of Huanghua pears (Pyrus pyrifolia Nakai, cv. Huanghua) treated with different kinds of coatings during storage. Postharvest Biol. Technol. 49: 171-179.
What is edible coating in bananas?
Edible coatings are biodegradable films made from materials like chitosan and gum arabic that slow ripening and extend shelf life.
Inhibition of Cell Wall Degrading Enzymes, chitosan gum arabic banana coating, edible coating banana shelf life, polygalacturonase inhibition in banana, banana postharvest physiology
