Journal of Environmental and Agricultural Sciences (JEAS). Jamil et al., 2019. Volume 19: 10-22
Open Access – Research Article
Comparative Study for Growth and Yield Performance of Oyster Mushroom (Pleurotus spp.) on Different Substrates under Temperate Condition
Faiza Jamil1, Abid Yaqoob1, Zerfishan Mehmood1, Abdul Hamid1, Zulfiqar Shah1, Muhammad Imtiaz2,
Nadia Jamil2, Raina Ijaz1,*
1Department of Horticulture, University of The Poonch Rawalakot, Azad Kashmir, Pakistan
2Soil and Environmental Biotechnology Division, National Institute for Biotechnology & Genetic Engineering, Faisalabad, Pakistan
Abstract: The farming of oyster mushroom is reported as an economically viable biotechnology process for conversion of different organic wastes. Due to the lack of know-how about the use of different local substrate for production of oyster mushroom, the work was carried out at Rawalakot, Azad Jammu and Kashmir, Pakistan with the main objective to assess the suitability of selected substrates, without using any supplements. Three different species of oyster mushroom i.e., P. sajor-caju, P. sapidus and P. erynjii, were spawned on easily available substrates (wheat straw, newspaper, pine needles) and their mixtures. These substrates were used as 100% wheat straw (T1), 100% newspaper (T2), 100% pine needles (T3), 50% wheat straw + 50% newspaper (T4), 50% newspaper + 50% pine needles (T5), 50% pine needles + 50% wheat straw (T6). Results illustrated that P. sapidus performed the best when grown on 100% newspaper (T2) in terms of days required for spawn run completion, pinhead’s appearance and maturation of fruiting bodies. But, maximum number of fruiting bodies, yield and biological efficiency was observed in P. sapidus when grown on 50% wheat straw+50% newspaper (T4). Maximum protein content was shown by P. sajor-caju and P. sajor-caju when grown on 100% pine needles (T3) and newspaper (T2), respectively. So, it was concluded that P. sapidus can be spawned successfully on newspaper substrate alone or in combination with any other lignocellulosic substrate for better yield of oyster mushroom under temperate climatic conditions.
Keywords: Mushroom, agro-watses, biological efficiency, fruiting body, growth, yield.
*Corresponding author: Raina Ijaz: rainaijaz@yahoo.com ________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
Cite this article as:
Jamil, F., A. Yaqoob, Z. Mehmood, A. Hamid, Z. Shah, M. Imtiaz, N. Jamil and R. Ijaz. 2019. Comparative study for growth and yield performance of oyster mushroom (Pleurotus spp.) on different substrates under temperate condition. Journal of Environmental & Agricultural Sciences. 19:10-22. [View Full-Text] [Citations] 
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
Copyright © Jamil et al., 2019. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium provided the original author and source are appropriately cited and credited.
Similar Articles Published in JEAS
- Özkan, H., M. Aasim. 2020. Comparison of in vitro regeneration potential of different preconditioned and nonconditioned explants of peanut (Arachis hypogaea L). Journal of Environmental & Agricultural Sciences. 22(2): 11-19. [Abstract] [Citations]
- Akbarzai, D.K., G.R. Akbari, L. Mohammadi, M.N. Saif and A. Farhang. 2022. AMMI analysis of yield stability and adaptability in barley: A case study of Afghanistan. Journal of Environmental & Agricultural Sciences. 24(1&2): 18-25. [Abstract] [View Full-Text] [Citations]
- Nasir, M.W., and Z. Toth. 2021. Effect of drought stress on morphology, yield, and chlorophyll concentration of Hungarian potato genotypes. Journal of Environmental & Agricultural Sciences. 23(3&4): 8-16. [View Full-Text] [Citations]
- Ali, I., M.T. Mahmood, M.I. Akhtar, A. Zafar, A. M. Khan M. Zubair and W. Anum. 2019. Some direct and indirect selection indices for increased yield of peas (Pisum sativum L.). Journal of Environmental& Agricultural Sciences. 21:23-28. [Abstract] [View Full-Text] [Citations]
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, Oyster Mushroom Cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
1. Introduction
Mushroom, a macro fungus which is eukaryotic, fleshy and spore bearing body typically epigeous or hypogenous (Chang and Wasser, 2017; Raghavendra et al., 2018). Out of the 1.5 million reported fungi species, it was estimated that 160,000 fungi species produce fruiting bodies of sufficient sizes and suitable structures to be considered as macro fungi (Hawksworth, 2012; Murugesan, 2017). These can be considered mushrooms according to the above definition. Additionally, of the 16,000 known mushroom species, about 700 are known to possess medicinal properties (Wasser, 2010). About 200 mushroom species are being used as functional foods around the world (Kalac, 2013) but only about 35 species are commercially cultivated (Aida et al., 2009; Xu et al, 2011).
The oyster mushrooms (Pleurotus spp.) rank third after the white button and shiitake among the world mushroom production (Gyorfi and Hajdu, 2007). Globally China is the major producer (~90% share) and consumer of oyster mushroom (Mohamed et al., 2016; Shah et al., 2004). During 2016, Pakistan earned a foreign exchange of 12.930 million dollars by exporting about 0.201 million kg of mushrooms (Razi, 2017).
Mushrooms are progressively becoming an important component of human’s diet globally (Ajonina and Tatah, 2012) because of their high nutritional value and medicinal properties (Chang and Miles, 2004). Mushrooms are famous worldwide as the vegetarian’s meat (Ahmed et al., 2009). It contains a high percentage of proteins, which is higher than any other vegetative protein, but it is low in calories (Alananbeh et al., 2014; Ananbeh, 2003). Having low starch content, it is a good diet for people suffering from diabetic (Devi et al., 2015). They contain 19-35% protein (Oei, 1991), 0.6-3.1% fat content (Anthony, 2007), 70-90% moisture content (Mckeller and Khorman, 1990), 7.5-16.5% fibre content (8.7 %), 9.8 % ash content and 57.6 % carbohydrate content, while vitamins such as riboflavin (4.7 mg) and niacin (108.7 mg), thiamin (4.8 mg), minerals like phosphorus (476 mg), ferrous (8.5 mg) calcium (98 mg) and sodium (61 mg) on 100 g dry weight basis, are also found in mushrooms (Pandey et al., 2000, Almomany and Ananbeh, 2011, Ananbeh and Almomany, 2005).
Substrate composition has significant impact on growth and nutritional composition of mushrooms (Bellettini et al., 2019; El Sebaaly et al., 2019). Mushroom potentially can be grown on different lignocellulosic materials like saw dust (Ananbeh, 2003; Hanafi et al., 2018), soybean straw, wheat straw (Cao et al., 2019) and groundnut shells (Mane et al., 2007), newspaper and tea leaves (Jain, 2005), cotton waste and paddy straw (Ashraf et al., 2013), date palm waste (Alananbeh et al., 2014). However, for rapid growth of mushrooms an ideal substrate should have nitrogen and carbohydrate contents, cellulose and lignin because of its efficient lignin degrading trait (Das and Mukherjee, 2007).
Annually wheat straw is produced in large quantity (3.2 to 11.0 million tons) in wheat base cropping system (Sarwar et al., 2002; Solovyev et al., 2018). This waste/by-product of wheat could be a cheap source for oyster mushroom cultivation. Paper is nearly 100% cellulosic in composition. Wastepaper refers to paper, newspaper and cardboard collected and discarded from industries, supermarkets and many other shopping places, residential places, and offices. At an international level, about 40-65% of paper is wasted and disposed of to the environment (Prognos, 2010) that is increasing pollution. Oyster mushroom cultivation could reduce this pollution at a safe level. Pine needles are widely available in the forests of Azad Kashmir. Pine needles has more than 50% holocellulose, and hence can be used for production of pulp and paper. It is also rich in lignin, which is a requirement of oyster mushroom cultivation (Priti et al., 2013; Ananbeh, 2003).
Despite the excellent climatic conditions, multiple cropping possibilities, low cost of production, abundance of man power and agro-waste resources, oyster mushroom is not being cultivated in most of the temperate climate conditions. This is mainly due to non-availability of suitable strains, lack of nutritional awareness and cultivation technology. So, keeping in view its easy cultivation practices, high nutritional value and profitable business to the growers, we aimed to evaluate the best variety and substrate of oyster mushroom to be used under temperate conditions.
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, Oyster Mushroom Cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficienc
2. Materials and Methods
2.1. Substrates and Spawn Preparation
The proposed research work was carried out at the Laboratory of the Department of Horticulture, Faculty of Agriculture, University of The Poonch Rawalakot, Azad Kashmir, Pakistan, during 2016-2017 for evaluation of three species of Pleurotus viz., P. sajor-caju, P. sapidus, P. erynjii, on three different substrates i.e., wheat straw, newspaper, pine needles and their mixtures. The spawn of these three species of oyster mushroom was taken from culture bank of Mushroom laboratory, Institute of Horticultural Sciences, University of Agriculture, Faisalabad. The detail of treatments plan is given in Table 1.
Wheat straw and newspaper were collected from local markets. Pine needles were collected from different locations around Rawalakot like Mutialmara, Maidan, and Jandali. The chopped substrates were soaked in water for 24 hours to moisten them thoroughly and were stalked on the steep cemented floor to remove the excessive moisture from the substrates to get 65-75% moisture level. The substrates were left under the sun for sterilization for 5 days by covering with polythene sheet before filling the bags. Each of the substrates and their combinations were filled in polypropylene bags of (8 × 12 cm) and their mouths were plugged by inserting water absorbing cotton with the help of plastic rings. Each of the six treatments was replicated three times. The bags were autoclaved at 121℃ at 15-20 lbs pressure and allowed to cool overnight.
2.2. Growth conditions for mushroom room
The room was provided with suitable climatic conditions for the proper growth of oyster mushroom. Room temperature and humidity were adjusted between 20-25 ℃ and 65-75%, respectively.
Mushroom cultivation has two important stages viz., spawn running and fructification, while temperature and humidity are two vital factors involved at both stages. The temperature was controlled by electric heaters at 25 ℃ for running of spawn and 17-20 ℃ for formation of fruiting body. Humidity was between 65-85% during spawn running and fructification. If the humidity of the bags was found less than optimum, it was achieved by spraying tab water on bags twice a day.
Exhaust fans were used for the exhaust of gases from mushroom growing room because oxygen is essential for mushroom during fructification. For this purpose, the pinholes were also made in the bags with the help of paper pins for exhaust of gases from bags. For proper fruiting, the bags were watered three times a day during cropping.
2.3. Morphological parameters
The data was collected, and observations were made on the following parameters: days for spawn run completion, days for appearance of pinheads, fruiting bodies maturation, number of fruiting bodies, yield and biological efficiency.
2.4. Chemical analysis
protein content estimation (%) was done through Kjeldahl method described by Chang and Buswell (2003) by using following equation [1].
[1]Where CP is crude protein (%) and N is nitrogen (%)
2.5. Statistical analysis
Data collected were analysed by using the analysis of variance (ANOVA) technique using M-STAT C software (Reference)); LSD test at 5% probability was applied to compare the difference among the treatments means (Steel et al., 1997).
3. Results
3.1. Days for completion of spawn run
Highly significant results were observed among treatments in terms of days taken for spawn running of three P. species (P. sajor-caju, P. sapidus and P. erynjii). These species behaved drastically on different growing media in completing their mycelial growth and treatment × variety interaction was highly significant.
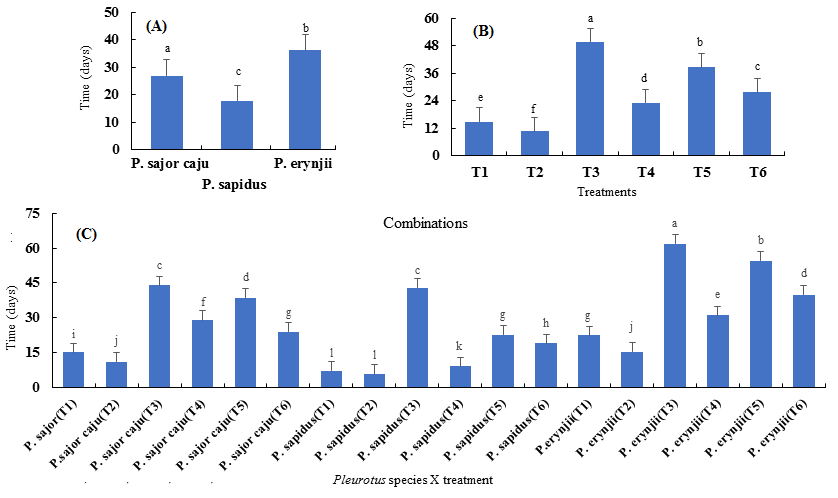
Among the three P. species, best results were obtained from P. sapidus, which needed minimum number of days (49.6) followed by P. sajor-caju with 67.6 days and P. eryngii was the least with 77.7 days for spawn run completion (Fig. 1 a). Among the six treatments, T2 was considered best because it needed minimum number of days (40.8) for spawn run completion. It was followed by T1, T4, T6, T5 and T3. Pine needles (T3) was considered the least, which took maximum number of days 87.556 days for spawn run completion (Fig. 1 b). Best interaction was observed for P. sapidus grown on newspaper (T2), which took 23.6 days for mycelial growth, whereas interaction between P. erynjii and pine needles (T3) required the maximum number of days (96.3) (Fig. 1 c).
http://jeas.agropublishers.com/wp-content/uploads/2026/03/Fig-1.bmp
Fig. 1. Means of the time (days) needed for spawn completion against species (A), treatments (B) and combinations (C) for the three Pleurotus spp.
3.2. Days for appearance of pinheads
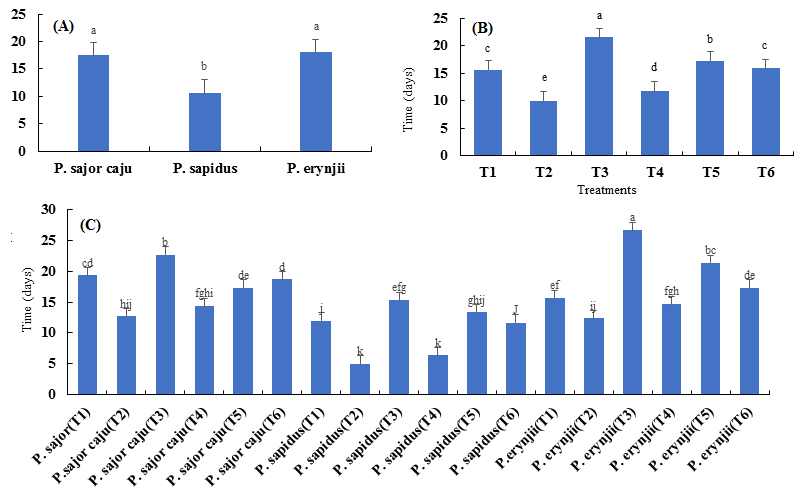
Highly significant results were observed among the three P. species in terms of days taken for pinhead appearance. Among the three species, best results were obtained from P. sapidus, which took minimum number of days (17.2), followed by P. sajor-caju with 26.9 days and P. eryngii was the least with 37.5 days (Fig. 2 a).
Fig. 2. Means of the time (days) needed for pinhead appearance of species (A), treatments (B) and combinations (C) for the three Pleurotus spp.
Among the six treatments, T2 was considered the best where it took minimum number of days (10.667) in terms of pinhead appearance. It was followed by T1, T4, T6, T5 and T3. Pine needles (T3) was considered the least where it took maximum number of days (49.6) for pinhead appearance (Fig. 2 b). Best interaction was observed by P. sapidus with newspaper (T2) and wheat straw (T1), which showed the same level of significance and took minimum number of days (5.6 and 5.7), respectively to approach pinhead appearance (Fig. 2 c).
3.3. Days for maturation of fruiting bodies
P. sajor-caju and P. erynjii were statistically similar in terms of days taken for pinhead appearance. Among the three species, best results were obtained from P. sapidus which took minimum number of days (10.6), followed by P. sajor-caju with 17.5 days and P. eryngii was the least with 18 days (Fig. 3 a). On the other hand, T2 needed minimum number of days (10.0) for fruiting bodies maturation, while pine needles (T3) took maximum number of days (21.5) for fruiting bodies maturation (Fig. 3 b). P. sapidus required minimum number of days when grown on newspaper (T2) and mixture of wheat straw and newspaper (T4), i.e. 5 and 6.3 days respectively; both had with same level of significance. While, maximum days were observed from P. erynjii (T3) with 26.6 days for fruiting bodies maturation (Fig. 3 c).
Fig. 3. Means of the time (days) needed for fruiting bodies maturation of the species (A), treatments (B) and combinations (C) for the three Pleurotus spp.
3.4. Number of fruiting bodies
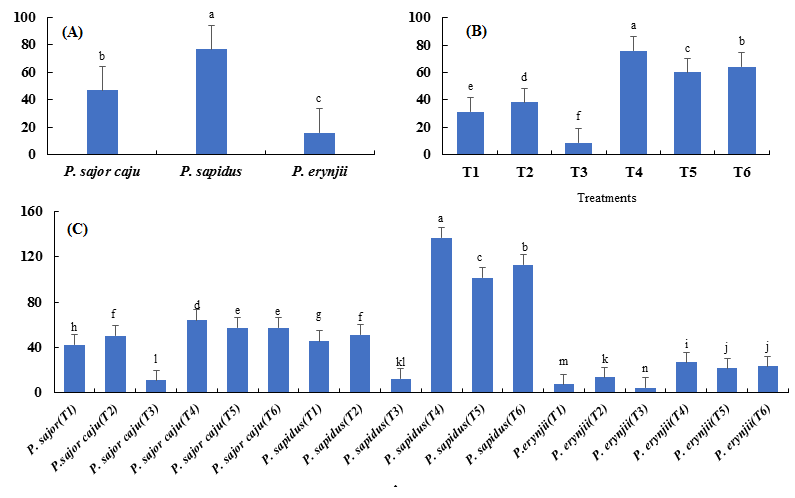
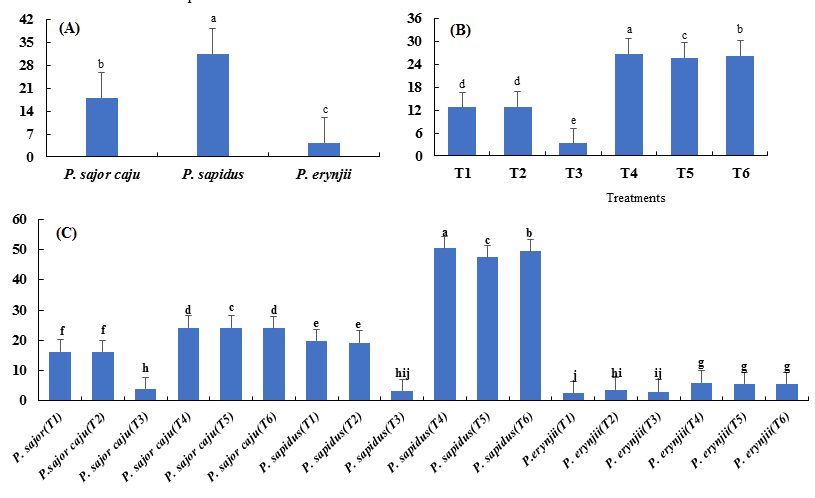
Among the three species, best results were obtained from P. sapidus, which produced maximum number of fruiting bodies (76.6) followed by P. sajor-caju with 46.7 fruiting bodies. P. eryngii was the least with minimum number of fruiting bodies (15.8) (Fig. 4 a). Among the six treatments, T4 showed the best results in terms of number of fruiting bodies. Least results were obtained from T3 (100 % pine needles) with 8.8 fruiting bodies (Fig. 4 b). P. sapidus grown on mixture of wheat straw and newspaper (T4) produced highest number of fruiting bodies (137). P. erynjii grown on pine needles (T3) was least with minimum number (4.0) of fruiting bodies (Fig. 4 c).
Fig. 4. Comparison of mean values of mushroom species against the species (A), treatments (B), and their combinations (C) for the number of fruiting bodies.
3.5. Total yield
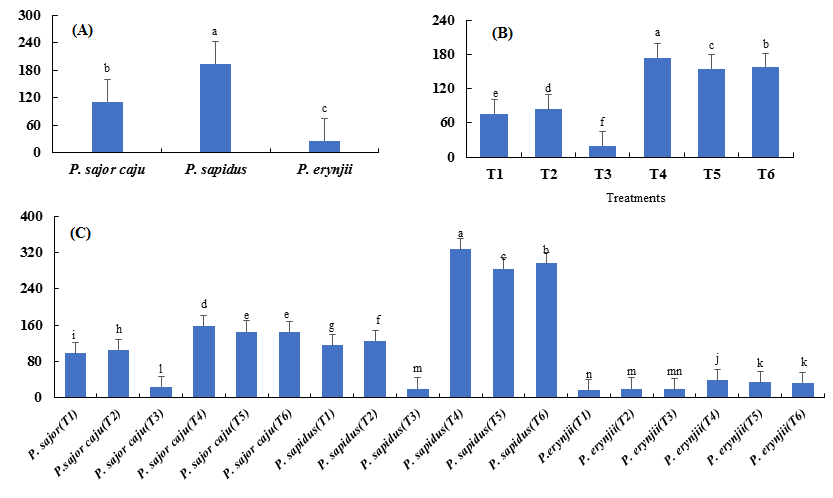
Highly significant results were observed among the three Pleurotus species in terms of total yield. Among the three species, best results were obtained for P. sapidus, which produced maximum total yield (194.83 g) followed by P. sajor-caju with 111.78 g while, P. eryngii had the least total yield (26.39 g) (Fig. 5 a). Among the six treatments, T4 (174.33 g total yield) was considered the best. Minimum mushroom yield (19.89 g) was produced from treatment T3 (100 % pine needles) (Fig. 5 b). Among interactions, P. sapidus (T4) showed the best interaction with a total yield of 328 g while, the lowest total yield (15 g) was recorded for P. erynjii (T1) (Fig. 5 c).
Fig. 5. Means of the total yield (percentage) of the species (A), treatments (B), and their combinations (C) for the three Pleurotus spp.
3.6. Biological efficiency
Highly significant results were observed among the three species in terms of biological efficiency. Best results were obtained from P. sapidus which showed maximum biological efficiency of 31.5 % followed by P. sajor-caju with 18.0 %. P. eryngii had the least biological efficiency (4.2 %) (Fig. 6 a). Among the six treatments, T4 was considered the best treatment with 26.8 % biological efficiency. Minimum results were obtained from T3 with biological efficiency of 3.293 % (Fig. 6 b). Among interaction, P. sapidus grown on mixture of wheat straw and newspaper (T4) showed the best interaction in terms of biological efficiency of 50.460 %. The lowest biological efficiency was recorded for P. erynjii (T3) with 2.5 % (Fig. 6 c).
Fig. 6. Means of the biological efficiency of the species (A), treatments (B), and their combinations (C) for the three Pleurotus spp.
3.7. Protein content
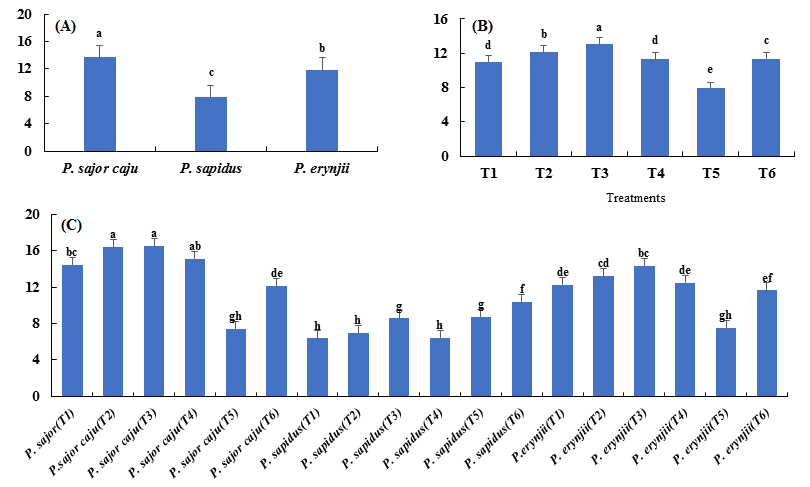
Among the three P. species, best results were obtained from P. sajor-caju which showed maximum protein content 13.668 % followed by P. erynjii with 11.904 % while P. sapidus had the least protein content (7.901 %) (Fig. 7 a).
Among the six treatments, T3 with 13.132 % protein content was considered the best followed by T2 with 12.169 %, T6 with 11.389 %, T4with 11.330 %, T1 with 11.021 % protein content. Minimum protein contents were obtained from T5 with 7.903 % (Fig. 7 b).
Fig 7. Means of the protein contents (percentage) of the species (A), treatments (B), and their combinations (C) for the three Pleurotus spp.
Among interactions, P. sajor-caju (T3) and P. sajor-caju (T2) showed the best interaction in terms of protein content with a value of 16.513 % and 16.387 % respectively. This result was followed by P. sajor-caju (T4), P. sajor-caju (T1), P. erynjii (T3),P. erynjii (T2), P. erynjii (T4), P. erynjii (T1), P. sajor-caju (T6), P. erynjii (T6), P. sapidus (T6), P. sapidus (T5), P. sapidus (T3), P. erynjii (T5), P. sajor-caju (T5), P. sapidus (T2), P. sapidus (T4) and P. sapidus (T1) with protein content of 15.103 %, 14.427 %, 14.320 %, 13.187 %, 12.460 %, 12.263 %, 12.130 %, 11.647 %, 10.390 %, 8.717 %, 8.563 %, 7.547 %, 7.447 %, 6.933 %, 6.427 % and 6.373 % respectively. The lowest protein content was recorded for P. Sapidus (T1) with a protein content of 6.373 % (Fig. 7 c).
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
4. Discussion
4.2. Days for completion of spawn run
P. sapidus needed minimum number of days followed by P. sajor-caju and P. eryngii was the least with maximum number of days for completing mycelial growth. This was due to green moulds attack which may be caused due to Trichoderma spp. or Penicillium spp. on some treatments during growth stage. This was experienced by Biswas and Kuiry (2013) who stated that competitor moulds attack (Aspergillus niger, Penicillium spp.) and insect’s infestation in the bags of Pleurotus spp. resulted in poor growth of mycelium which contributed negatively towards spawn run and yield.
Peksen and Kucukomuzla, (2004) studied the spawn run completion of P. sajor-caju and P. sapidus on various substrates and reported that P. sapidus took minimum number of days for spawn run completion than P. sajor-caju which has a resemblance to our finding. P. sapidus (T2) took 23 days to complete mycelial growth whereas P. erynjii (T3) took 96 days. P. erynjii is more susceptible to diseases and more sensitive to growing environments (Kirbag and Akyuz, 2008). Baysal et al. (2003) findings completely agree with the finding of this experiment that newspaper as the sole or combined with supplemented substrate is suitable for oyster mushroom production. Oei, (1991) stated that mycelial growth period of all P. species varies within the range of 2-8 weeks depending upon the structure of substrates and species.
Croan, (2004) cultivated different P. species on untreated pine chips. These fungi did not grow or grew poorly on untreated pines. He stated that it may be due to the reason that pine extractives were poisonous to several wood-residing fungi. The same explanation regarding pine needles was also stated by Alananbeh (2003) on P. osteratus.
4.3. Days for appearance of pinheads
P. sapidus took minimum number of days followed by P. sajor-caju and P. eryngii. Mshandete, (2011) observed that P. sapidus showed pinhead appearance in 8-13 weeks on different substrates used. This has some resemblance with our findings. The result of P. erynjii has a resemblance with the findings of Kirbag and Akyuz, (2008) that the time needed for pinhead appearance of P. erynjii was 26.22 to 44.2 days depending upon the substrate type used. T2 was considered the best with minimum number of days in terms of pinhead appearance whereas T3 was considered least where it took maximum number of days.
This research has some resemblance with the findings of Baysal et al. (2003). They stated that pinhead appearance after spawn running in sole paper substrate appeared within 6-7 days. Also, some researchers stated that mushrooms are observed to be successfully grown on different agricultural residues which are rich in carbon sources (lignin, cellulose and hemicellulose) such as leaves of banana, cereal straw, newspaper, sawdust, date palm leaves and droppings of poultry (Onuoha. 2007;Fasidi and Kadiri, 1993, Alananbeh et al., 2014; Ananbeh, 2003, Ananbeh and Al-Momany, 2005; Almomany and Ananbeh, 2011).
This is also supported by other Pleurotus investigators such as Khan et al. (2001) that pinhead formation took 7-8 days in various substrates. Whereas interaction between P. erynjii (T3) was not satisfactory with maximum number of days (62) for pinhead appearance. This may be due to the high quality of cellulose and lignin contents which needed longer time to start pinning and fruiting body formation (Oei, 2003). Lignin and cellulose contents of pines were reported by (Pereira, 1998) to be 3-29 and 42-49 respectively which are higher than newspaper and wheat straw.
4.4. Days for maturation of fruiting bodies
P. sapidus took minimum number of days 10.611, followed by P. sajor-caju with 17.5 days and P. eryngii was the least with 18 days. Mshandete, (2011) stated that it took 2-4 days to develop to harvestable mushrooms by P. sapidus. But this experiment has value more than that of (Mshandete, 2011) findings. This may be due to temperature fluctuations during fruiting period.
Fruiting bodies maturation completed earlier in T2 (10 days) and late in T3 (21 days). Baysal et al. (2003) reported that fruiting bodies maturation occurs within five days for 100 % paper substrate. But in this experiment fruiting bodies maturity on treatment T2 (100 % newspaper) was within 10 days which is greater than the days of Baysal’s findings but having some resemblance. In this aspect, other results came-up with different results regarding to the maturity periods of species of oyster mushroom. (Khan and Ali, 1981) observed a maturity period between 21 to 28 days using seed of cotton as substrate. According to Khanna and Garcha (1981) about 104 days may be taken up by the oyster mushroom to give harvestable mushrooms on paddy straw. That is similar with this experiment in which the substrate (100% pine needles) spawned by P. erynjii took more than 100 days to complete its fruiting bodies maturity. These variations in harvesting periods may come from the differences in the growing conditions (controlled or semi-controlled conditions) and physiological factors for mushroom cultivation. For instance, the constant temperature, humidity and light arrangements (Girmay et al., 2016).
4.5. Number of fruiting bodies
Best results were obtained from P. sapidus which produced maximum number of fruiting bodies followed by P. sajor-caju and P. eryngii was the least with minimum number of fruiting bodies. Moonmoon et al. (2010) obtained small number of fruiting bodies from the king oyster mushroom. Kopinski and Marks (2012) stated that the substrate composed of newspaper alone produced net fruiting. It was contrary to the findings of this experiment.
4.6. Total yield
Best results were obtained from P. sapidus which produced maximum total yield (194.83 g) followed by P. sajor-caju while P. eryngii had the least total yield. Similarly, Dundar et al. (2008) obtained lowest yield (4.5 g) from P. eryngii. T4 was considered best and minimum results were obtained from T3. This study has also a resemblance with the findings of Kopinski and Marks (2012). They found that maximum yield was observed in newspaper and wheat straw in the ratio of 3:1 respectively. Similarly, Yildiz et al. (2002) stated that the mixtures which included the paper generally produced higher yields. Obodai et al. (2003) stated that yield obtained from different lignocellulosic substrates were 183.1 g, 151.8 g, 111.5 g, 87.8 g, 49.5 g, 23.3 g, 13.0 g and 0.0 g which clearly matches to this experiment finding.
Balasubramanya and Kathe (1996) stated that the fungus Penicillium spp. and Trichoderma spp. competed with the species of Pleurotus after pasteurisation with hot water (80°C for 2h) may be due to the fractional breakdown of cellulose and hemicellulose thus making them accessible to competitors. This was also experienced by the treatment (100% pine needles) of this experiment which was rapidly attacked by Trichoderma spp. due to which it gives lowest yield. Pokhrel et al. (2013) stated that poor yield performance by substrates may be due to the reason that lignin and cellulose took longer or poorly to break down as in the case of yield of P. erynjii (T1) in this experiment.
4.7. Biological efficiency
P. sapidus showed maximum biological efficiency of 31.597 % followed by P. sajor-caju and P. eryngii had the least biological efficiency. The result of P. erynjii is contrary to the findings of Moonmoon et al. (2010). They observed highest biological efficiency from P. erynjii on saw dust and rice straw than other strains. Croan, (2004) cultivated P. sajor-caju, P. sapidus and P. erynjii on pines and obtained the biological efficiency of 131, 154 and 139 % respectively which is contrary to this experiment finding. Wang et al. (2001) and Rizki and Tamai (2011) used several substrates to cultivate Pleurotus spp. and the biological efficiency values varied from 17 to 79 %. These observations agree with the findings of this experiment in terms of biological efficiency. Kalita et al. (1997) observed biological efficiency (BE) for various lignocellulosic materials ranged from 35.2-60.9 %. The lower biological efficiency may be due to the fact that the higher mushroom yield and biological efficiency correspond to the mycelia growth, colonization period and harvest period (Pokhrel et al., 2013).
4.8. Protein content
P. sajor-caju showed maximum protein content 13.668 % followed by P. erynjii while P. sapidus had the least protein content. Khan et al. (2008) stated that maximum protein was found in P. sajor-caju 24.5 % which was followed by P. erynjii (21.9 g/100g of dry weight) cultivated on different substrates. According to other Pleurotus investigators Breene (1990), Çokuner and Özdemir (2000), mushrooms contain protein ranging from 19 to 39 % in 100 g dried matter which is contrary to the findings of this experiment. Mandhare et al. (2008) found protein content to be lower in P. saqpidus as compared to other species used for the cultivation and also said that the nutritional value of the P. spp. varies from species to species when cultivated on one substrate.
The variation in the reported nutritional analysis in different Pleurotus spp. is due to several factors such as strains, type of substrate, spawning rate and media (Stamets, 1993). Singh and Kumar (2012) also stated that Pleurotus species have the ability to fix nitrogen from air. The difference in crude protein might be due to the differences in nitrogen contents of growth substrates.
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
5. Conclusion
Commercial production of oyster mushrooms mainly depends on the availability of cheap agricultural lingo-cellulosic residues which represents the ideal and most promising substrates for cultivation. The substrates used in this research can be considered practical and economically feasible under the temperate climatic conditions due to their availability throughout the year at little or no cost in large quantities. Additionally, the lignocellulosic agricultural residues like pine needles that are usually incorporated in soil, burned on the fields or left to rot in temperate zone could be a better and cheaper source for oyster mushroom cultivation and this can provide an economical gain to the people. Moreover, the availability of protein rich mushrooms could be made possible.So, the results showed that P. sapidus can be spawned successfully on newspaper substrate alone or in combination with any other lignocellulosic substrate for oyster mushroom cultivation under temperate environment and the maximum yield can be obtained from this substrate. Further research could be done on pine needles substrate to make it high yielding for farmers under temperate conditions as it is a very cheap and easily available substrate in the said area.
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
List of Abbreviations: CRD: Completely randomize design; CP: Crude protein; P: Pleurotus; S: Substrate; WS: Wheat straw; NP: Newspaper; PN: Pine needles; BE: Biological efficiency; gkg-1 : gram per kilogram; ANOVA : Analysis of variance; LSD: least significant difference.
Competing Interest Statement: All the Authors declare that they have no competing interest.
Author’s Contribution: FJ and RI conceived and designed of the study. FJ, AY and ZM conducted the experiment and FJ, AH and ZH performed the data analysis. FJ, MI and NJ wrote the manuscript. Finally, RI approved the current version of manuscript. All the authors read and approved the final manuscript.
Acknowledgments: This study was conducted without funding. Authors are thankful to Dr. Shahid Iqbal Awan, The Poonch Rawalakot, Azad Kashmir, Pakistan for his interest, skills and rousing guidance during this study.
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, Oyster Mushroom Cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
References
Ahmed, S.A., J.A. Kadam, V.P. Mane, S.S. Patil and M.M.V. Baig. 2009. Biological efficiency and nutritional contents of Pleurotus florida (Mont.) singer cultivated on different agro-wastes. Nat. Sci. 7(1):44.
Aida, F.M.N.A., M. Shuhaimi, M. Yazid and A.G. Maaruf. 2009. Mushroom a s a potential source of prebiotics: a review. Trends Food Sci. Technol. 20: 567-575.
Ajonina, A.S and L.E. Tatah. 2012. Growth performance and yield of oyster mushroom (Pleurotus ostreatus) on different substrates composition in Buea South West. J. Biochem.3: 139-145.
Alananbeh, K.M., N.A. Bouqellah and N.S. Al Kaff. 2014. Cultivation of oyster mushroom Pleurotus ostreatus on date-palm leaves mixed with other agro-wastes in Saudi Arabia. Saudi J. Biol. Sci. 21(6): 616–625.
Al-Momany, A and K. Ananbeh. 2011. Conversion of agricultural wastes into value added product with high protein content by growing Pleurotus ostreatus. Environ. Earth Sci. 9:1483-1490.
Ananbeh, K.M and A.R. Almomany. 2005. Production of oyster mushroom Pleurotus ostreatus on olive cake agro waste. Dirasat, Agric. Sci. 32(1): 64-70.
Ananbeh, K.M. 2003. Production of oyster mushroom on different agricultural wastes available in Jordan. Thesis, University of Jordan, Jordan.
Anthony, M.M. 2007. Proximate and nutrient composition of three types of indigenous edible wild mushrooms grown in Tanzania and their utilization prospects. Afr. J. Food Agric. Nutr. Dev. 7: 2-9.
Ashraf, J., M.A. Ali, W. Ahmad, C.M. Ayyub and J. Shafi. 2013. Effect of different substrate supplements on oyster mushroom (Pleurotus spp.) production. Food Sci. Technol.1(3): 44-51.
Balasubramanya, R.H and A.A. Kathe. 1996. An inexpensive pre-treatment of cellulosic materials for growing edible oyster mushrooms. Biol. Res. Technol. 57(3): 303-305.
Baysal, E., H. Peker, M.K. Yalinkilic and A. Temiz. 2003. Cultivation of oyster mushroom on waste paper with some added supplementary materials. Bioresour. Technol. 89: 95–97.
Bellettini, M.B., F.A. Fiorda, H. A. Maieves, G.L. Teixeira, S. Avila, P.S. Hornung, A. M. Junior and R.H. Ribani. 2019. Factors affecting mushroom Pleurotus spp. Saudi J. Biol. Sci. 26(4): 633-646.
Breene, W.M. 1990. Nutritional and medicinal value of specialty mushrooms. J. Food Prot. 53(10): 883-894.
Cao, G., T. Song, Y. Shen, Q. Jin, W. Feng, L. Fan and W. Cai. 2019. Diversity of bacterial and fungal munities in wheat straw compost for Agaricus bisporus cultivation. Hort. Sci. 54: 100-109.
Chang, S.T and P.G. Miles. 2004. Mushrooms: Cultivation, nutritional value, medicinal effect and environmental impact. CRC Press, Boca Raton. p.415.
Chang, S.T. and S.P. Wasser. 2017. The Cultivation and Environmental Impact of Mushrooms. Oxford University Press. Oxford Research Encyclopedia of Environmental Science. Retrieved 18 Jun. 2019, from https://oxfordre.com/environmentalscience/view/10.1093/acrefore/9780199389414.001.0001/acrefore-9780199389414-e-231.
Çokuner, Y and Y. Özdemir. 2000. Acid and EDTA blanching effects on the essential element content of mushrooms (Agaricus bisporus). J. Sci. Food Agr. 80: 2074-2076.
Croan, S.C. 2004. Conversion of conifer wastes into edible and medicinal mushrooms. J. For Prod. 54(6): 68-76.
Das, N. and M. Mukherjee. 2007. Cultivation of Pleurotus ostreatus on weed plants. Bioresour. Technol. 98(14): 2723-2726.
Devi, M.K., B. Haobam, T.I. Devi1, W.N. Singh and W.R. Singh. 2015. Study of spawning response of Pleurotus sajor-caju in two different substrates (wheat and rice). J. Sci. Eng. Res. 6: 1-3.
Dundar, A., H. Acay and A. Yildiz. 2008. Yield performance and nutritional contents of three oyster mushroom species cultivated on wheat stalk. Afr. J. Biotechnol. 7(19): 3497-3501.
El Sebaaly, Z., F. Assadi, Y.N. Sassine and N. Shaban. 2019. Substrate types effect on nutritional composition of button mushroom (Agaricus bisporus). Agric. Forest. 65(1): 73-80
Fasidi, I.O and M. kadiri. 1993. Use of agricultural wastes for the cultivation of Lentinus subnudus in Nigeria. Rev. Biol. Trop. 41: 41-45.
Girmay, Z., W. Gorems, G. Birhanu and S. Zewdie. 2016. Growth and yield performance of Pleurotus ostreatus (Jacq. Fr.) Kumm (oyster mushroom) on different substrates. Appl.Indust.Microbiol. Technol. 6(1): 87.
Gyorfi, J. and C.S. Hajdu. 2007. Casing-material experiments with Pleurotus eryngii. Int. J. Hort. Sci. 13: 33-36.
Hanafi, F.H.M., S. Rezania, S.M. Taib, M.F.M. Din, M. Yamauchi, M. Sakamoto, H. Hara, J. Park and S.S. Ebrahimi. 2018. Environmentally sustainable applications of agro-based spent mushroom substrate (SMS): an overview. J. Mater. Cycles Waste Manag. 20(3): 1383-1396.
Hanafi, F.H.M., S. Rezania, S.M. Taib, M.F.M. Din, M. Yamauchi, M. Sakamoto, H. Hara, J. Park, S.S. Ebrahimi. 2018. Environmentally sustainable applications of agro-based spent mushroom substrate (SMS): an overview. J. Material Cycles Waste Management. 20(3): 1383-1396.
Hawksworth, D.L. 2012. Global species numbers of fungi: Are tropical studies and molecular approaches contributing to more robust estimate? Biodivers. Conserv. 21: 2425-2433.
Jain, A.K. 2005. Studies on cultivation technology of Pleurotus spp. and their potential in Sagar region (Ph.D. thesis).
Kalac, P. 2013. A review of chemical composition and nutritional value of wild-growing and cultivated mushrooms. J. Sci. Food Agric. 93: 209-218.
Kalita, P., N. Mazumder and P. Kalita. 1997. Performance of oyster mushroom on certain plant wastes. Indian J. Agric. Sci. 14: 221-224.
Khan, A.M., S.M. Khan and A.S. Shakir. 2001. Studies on the cultivation of the oyster mushroom on different substrates. Pak. J. Phytopathol. 13: 140-143.
Khan, M.A., S.M.R. Amin, M.N. Uddin, M. Tania and N. Alam. 2008. Comparative study of the nutritional composition of oyster mushrooms cultivated in Bangladesh. Bangladesh J. Mushroom. 2(1): 9-14.
Khan, S.M and M.A. Ali. 1981. Cultivation of oyster mushroom (Pleurotus spp.) on cotton boll locules. Mushroom Sci. 11: 691-692.
Khanna, P and H. C. Garcha. 1981. Introducing the cultivation of Pleurotus florida in the plains of India. Mushroom Sci. 11: 655-665.
Kirbag, S and M. Akyuz. 2008. Effect of various agro-residues on growing periods, yield biological efficiency of Pleurotus eryngii. J. Food, Agric. Environ. 6(3-4): 402-405.
Kopinski, L and S.K. Marks. 2012. Utilization of waste newspaper using oyster mushroom mycelium. Ind. Eng. Chem. Res. 51: 4440-4444.
Mandhare, V.K., A.V. Suryawanshi and S.N. Mate. 2008. Productivity and nutritive value of Pleurotus spp. on wheat straw and its effects on nutritional indices of spent straw. Agric. Sci. Dig. 28(1): 1-5.
Mane, V.P., S.S. Patil, A.A. Syed and M.M.V. Baig. 2007. Bioconversion of low quality lignocellulosic agricultural waste into edible protein by Pleurotus sajor-caju (Fr.) Singer. J. Zhejiang Uni. Sci. 8(10): 745-751.
Mckeller, R.L. and R.E. Kohrman. 1990. Oyster mushroom production. J. Agric. Food Chem. 24: 549-570.
Mohamed, M.F., E.F.S. Refaei, M.M.A. Abdalla and S.H. Abdelgalil. 2016. Fruiting bodies yield of oyster mushroom (Pleurotus columbinus) as affected by different portions of compost in the substrate. Int. J. Recycl. Org. Waste Agric. 5(4): 281-288.
Moonmoon, M., M.N. Uddin, S. Ahmed, N.J. Shelly and M.A. Khan. 2010. Cultivation of different strains of king oyster mushroom (Pleurotus eryngii) on saw dust and rice straw in Bangladesh. Saudi J. Biol. Sci. 17(4): 341-345.
Mshandete, A.M. 2011. Cultivation of Pleurotus HK-37 and Pleurotus sapidus on cattail (Typha domingesis) substrate in Tanzania. Int. J. Res. Biol. Sci. 1(3): 35-44.
Obodai, M. and K.A. Vowotor. 2002. Performance of various strains of Pleurotus spp. under Ghanaian conditions. J. Food Technol. Africa. 3: 878-968.
Obodai, M., J.C. Okine and K.A. Vowotor. 2003. Comparative study on the growth and yield of Pleurotus ostreatus mushroom on different lignocellulosic by-products. J. Ind. Microbiol. Biotechnol. 30(3): 146-149.
Oei, P. 1991. Cultivation on fermented substrate. Manual on mushroom cultivation. Tool Publications, Netherlands, 249 pp.
Oei, P. 2003. Mushroom cultivation, appropriate technology for mushroom growers. Backhugs Publishers, Leiden. The Netherlands.
Onuoha, C. I. 2007. Cultivation of the mushroom (Pleurotus tuber regium) using some local substrates. Life Sci. J. 4(4): 58-61.
________________________________________________________________________________________________
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency
________________________________________________________________________________________________
Pandey, A., C.R. Soccol, P. Nigam, D. Brand, R. Mohan and S. Roussos. 2000. Biotechnological potential of coffee pulp and coffee husk for bioprocesses. J. Biochem. Eng. 6(2): 153-162.
Peksen, A and B. Kucukomuzlu. 2004. Yield potential and quality of some Pleurotus spp. grown in substrates containing hazelnut husk. Pak. J. Biol. Sci. 7: 768-771.
Pereira, H. 1998. Variability in the chemical composition of plantation eucalyptus. Wood Fiber Sci. 20(1): 82-90.
Pokhrel, C.P., N. Kalyan, U. Budathoki and R.K.P. Yadav. 2013. Cultivation of Pleurotus sajor-caju using different agricultural residues. Int. J. Agric. Policy Res. 1(2): 019-023.
Priti, S.L., A. Sharma and V. Bist. 2013. Pine needle – An evaluation of pulp and paper making potential. J. For. Prod. Ind. 2(3): 42-47.
Prognos, J. 2010. Use of agricultural waste materials in the cultivation of mushrooms. Mushroom Sci. 15: 3-23.
Raghavendra, V.B., C. Venkitasamy, Z. Pan and C. Nayak. 2018. Functional Foods from Mushroom. In: Gupta, V.K., Treichel, H., Shapaval, V.O., de Oliveira, L.A., Tuohy, M.G. (Eds.), Microbial Functional Foods and Nutraceuticals. John Wiley & Sons Ltd, pp. 65-91.
Razi, S. 2017. Pakistan exports 201,000kg mushrooms worth $12.930m in 2016. Daily Times, 03 January 2017.
Rizki, M and Y. Tamai. 2011. Effects of different nitrogen rich substrates and their combination on the yield yield performance of oyster mushroom (Pleurotus ostreatus). World J. Microbiol. Biotechnol. 27(1):1695-1702.
Sarwar, M., M.A. Khan and Z. Iqbal. 2002. Feed Resources for livestock in Pakistan. Int. J. Agric Biol. 4(1): 186-92.
Shah, Z.A., M. Ashraf and C. Ishtiq. 2004. Comparative study on cultivation and yield performance of oyster mushroom (Pleurotus ostreatus) on different substrates (Wheat straw, Leaves, saw dust). Pakistan J. Nutr. 3(3): 158-160.
Singh, M.P and V. Kumar. 2012. Biodegradation of vegetable and agro wastes by Pleurotus sapidus: A noble strategy to produce mushroom with enhanced yield and nutrition. Cell. Mol. Biol. 58(1): 1-7.
Solovyev, N., N.T. Prakash, P. Bhatia, R. Prakash, E. Drobyshev, B. Michalke. 2018. Selenium-rich mushrooms cultivation on a wheat straw substrate from seleniferous area in Punjab, India. J. Trace Elem. Med.e Biol. 50: 362-366.
Stamet, P. 1993. The Oyster mushroom of the genus Pleurotus. Growing Gourmet Med. Mushrooms 2: 283-284.
Steel, R.G.D., J.H. Torrie and D.A. Dickey. 1997. Principles and procedures of Statistic: a biometrical approach. 3rd Edition. McGraw-Hill Publishing Co. Boston, Massachusetts, 635 pp.
Wang, D., A. Sakoda and M. Suzuki. 2001. Biological efficiency and nutritional value of Pleurotus ostreatus cultivated on spent beer grain. Bioresour. Technol. 78(3): 293-300.
Wasser, S.P. 2010. Medicinal mushroom science: History, current status, future trends, and unsolved problems. Int. J. Med. Mushrooms, 12: 1-16.
Xu, X., H. Yan, J. Chen and X. Zhang. 2011. Bioactive proteins from mushrooms. Biotechnolo. Adv. 29(6): 667-674.
Yildiz, S., U.C. Yildiz, E.D. Gezer and A. Temiz. 2002. Some lignocellulosic wastes used as raw material in cultivation of the Pleurotus ostreatus culture mushroom. Process Biochem. 38(3): 301-306.
Growth and Yield Performance of Oyster Mushroom, oyster mushroom cultivation, Pleurotus spp. growth and yield, mushroom substrate comparison, oyster mushroom yield performance, mushroom biological efficiency